Fat Composition Affects T Cell-Mediated Immunity
- The fat content in cells controls their response to iron.
- Ferroptosis, a form of cellular death that is mediated by iron, occurs in T cells and is triggered by the oxidation of polyunsaturated fatty acids.
- More of these kinds of acids in the diet mean that ferroptosis is more likely to occur.
- In mice, CAR T anti-leukemia therapies were found to be more effective in the presence of more polyunsaturated fatty acids.
Scientists have found that the ratio between poly- and monounsaturated fatty acids affects the viability of T cells as well as both humoral and anti-tumor immunity [1].
The iron death
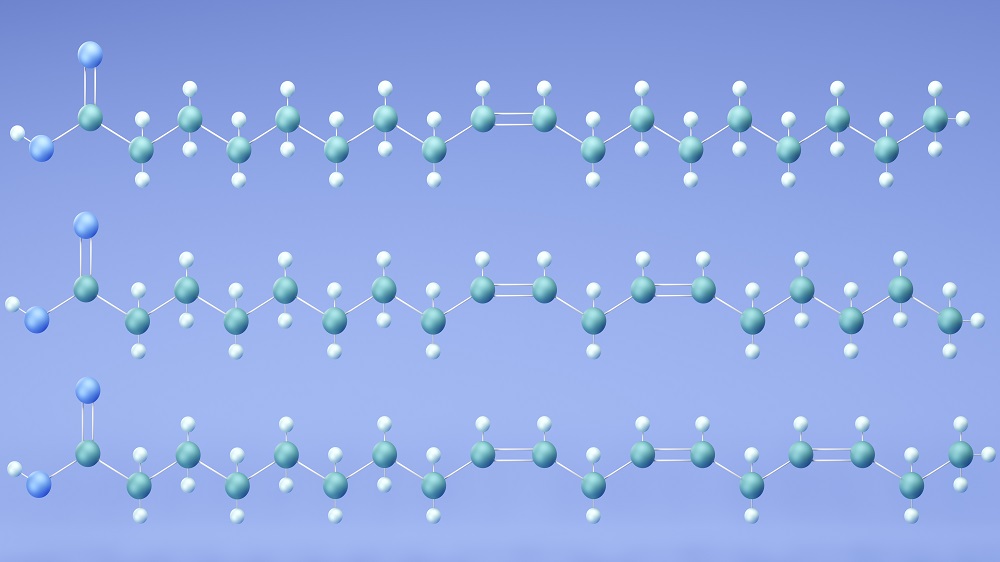
Some fats are broadly recognized as healthy while others are considered unhealthy, but the reality might be more complicated than that. Poly- and monounsaturated fats (PUFAs and MUFAs) are both considered to be healthy. PUFAs are abundant in fatty fish and nuts, while MUFAs are in olive oil and avocado. However, a new study from the University of Queensland and international collaborators, published in Nature, suggests that, when it comes to immunity, PUFAs and MUFAs are far from equal.
Apart from the usual form of programmed cell death, apoptosis, T cells are also regulated by a different iron-mediated death pathway called ferroptosis [2]. In ferroptosis, PUFAs, which have multiple double bonds, sit in phospholipids, the main structural element of cellular membranes. Iron-dependent chemistry oxidizes those double bonds in a chain reaction, generating toxic lipid hydroperoxides that perforate the membrane.
Prior work had established that ferroptosis regulates anti-tumor immunity, antibody-related (humoral) immunity, and memory T cell longevity [3]. A fundamental unanswered question was: what determines how resistant a given T cell is to ferroptosis in the first place?
The authors stumbled onto a clue from an unexpected source: they noticed that T cells from mice fed different standard laboratory diets (all considered nutritionally complete) had dramatically different susceptibility to ferroptosis. This observation was striking because these diets are used interchangeably across the field, with the implicit assumption that they are functionally equivalent for immune studies. So, the team dug deeper.
Establishing the mechanism
First, the researchers put young mice on one of the three standard rodent diets, SF-NIH31, SF00-100, or SF-AIN93G, which have high, medium, and low PUFA/MUFA ratios, respectively. All groups grew normally with comparable splenic T cell numbers and subset distributions. Splenocytes were then challenged ex vivo with increasing concentrations of a GPX4 inhibitor. GPX4 is the cell’s main defense against ferroptosis, which neutralizes hydroperoxides before they become lethal. The concentration needed to kill 50% of cells was defined as the “ferroptosis resistance index” (FRI).
T cells from high-ratio-fed mice were the most sensitive to ferroptosis, with FRIs two- to threefold lower than those from low-ratio-fed animals. This was confirmed with an alternative ferroptosis inducer. A ferroptosis inhibitor rescued all death, confirming it was genuinely ferroptotic. The entire effect was ferroptosis-specific: inducing apoptosis worked equally across the cells.
To test the effect in living animals, mice with T cell-specific GPX4 deletion, which makes them vulnerable to ferroptosis, were fed each diet. The severity of T cell loss in these mice was dramatically diet-dependent. On the low-ratio diet, the animals retained 35% of CD8+ T cells relative to controls – 3.5-fold more than on the high-ratio diet. CD4+ T cells showed a similar but milder pattern.
The next question was: does this actually matter for an immune response? The team chose to test this using TFH cells – a specialized subset of T cells that help B cells produce high-quality antibodies. Previous work had shown TFH cells are especially vulnerable to ferroptosis, making them a sensitive readout. The recipient mice were unable to form their own TFH cells, so, any antibody response had to come from the transferred T cells.
The results were stark: on the low-ratio diet, these GPX4-deficient T cells produced almost normal levels of TFH cells, several times higher than on the high-ratio diet. The authors systematically excluded alternative explanations, including gene expression, microbiota, and individual nutrients, and directly confirmed that FRIs inversely correlated with the PUFA/MUFA ratio across all three diets.
The PUFA/MUFA ratio in T cell zones, the areas where T cells preferentially reside, mirrored the dietary ratio. Crucially, free fatty acid abundance in T cell zones was correlated with corresponding phospholipid species. The researchers concluded that dietary PUFAs arrive in lymphoid tissue, get incorporated into T cell membrane phospholipids, and create more substrate for lipid peroxidation.
Superior anti-tumor immunity
Having established DEFs for humoral immunity, the team moved to anti-cancer immunity. In mice inoculated with melanoma, tumors that trigger adaptive immunity grew significantly slower in low-ratio-fed mice. These mice also had more effector CD8+ T cells and dramatically greater clonal expansion.
The researchers did not stop there and performed experiments in humans. In 24 healthy young adults, plasma PUFA/MUFA ratios in phospholipids were positively correlated with T cell lipid ROS and negatively correlated with T cell FRI. A parallel cohort of 12 children showed the same pattern. Notably, BMI itself did not correlate with FRI in either cohort; lipid composition, not body mass, predicted ferroptosis vulnerability.
Going back to animal experiments, immunodeficient mice were engrafted with human leukemia cells and treated with human CAR T cells. In low-ratio-fed mice, these cells had lower lipid ROS and two- to fourfold greater persistence in circulation. Without CAR T therapy, leukemia progressed identically on both diets, but with CAR T therapy, low-ratio-fed mice achieved markedly better tumor control and survival.
“The findings mark a profound leap in understanding of how our diet directly impacts immune system function,” said Professor Di Yu of UQ’s Frazer Institute. “Our immune system relies on T cells to manage the body’s immune response. The kinds of fats you eat change the fat composition inside your T cells, and those changes can make T cells either weaker or stronger in terms of immune protection. This discovery shows that dietary changes could potentially boost the effectiveness of vaccines and cancer therapies.”
Literature
[1] Wang, N., Chen, Z., Yao, Y., Sun, C., Wei, W., Sun, L., Zhang, H., Li, F., Butcher, D., Sun, S. R., Gong, J., Jiang, Y. C., Qi, Y., Huang, J., Nettelfield, S., Liu, R., Zheng, X., Li, C., Fu, Y., Geng, H., … Yu, D. (2026). Lipid metabolism drives dietary effects on T cell ferroptosis and immunity. Nature, 10.1038/s41586-026-10193-4. Advance online publication.
[2] Matsushita, M., Freigang, S., Schneider, C., Conrad, M., Bornkamm, G. W., & Kopf, M. (2015). T cell lipid peroxidation induces ferroptosis and prevents immunity to infection. Journal of Experimental Medicine, 212(4), 555-568.
[3] Ma, X., Xiao, L., Liu, L., Ye, L., Su, P., Bi, E., … & Yi, Q. (2021). CD36-mediated ferroptosis dampens intratumoral CD8+ T cell effector function and impairs their antitumor ability. Cell metabolism, 33(5), 1001-1012.