Michael Rae is the author of seven scientific articles in peer-reviewed journals as well as four book chapters in academic volumes. He is a long-time member and one-time Board Member of the Calorie Restriction Society and its Vice President for Research from 2014.
Related News
September 17, 2025
[Mountain View, September 17, 2025] — Lifespan Research Institute (LRI) today announced the launch of the Public Longevity Group (PLG), a new initiative focused on bridging the cultural gap between scientific breakthroughs in aging and their public acceptance. To kickstart its work, PLG has opened a crowdfunding campaign to develop tools that measure and strengthen...
July 27, 2021
We recently caught up with Dr. Aubrey de Grey and Michael Rae from SENS Research Foundation (SRF) about the landscape in the field. We asked them two simple questions, and they joined forces to give us their combined insights. lifespan.io: First, do you think we can defeat aging? Of course I think we can defeat...
Articles from this author
December 05, 2024
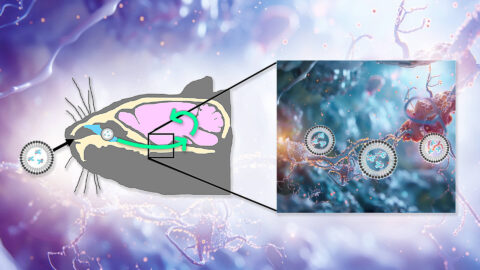
Aberrant tau inside neurons is a key driver of Alzheimer’s disease, but nearly all the therapies in development to target it can only capture the small amount that floats outside of them. A new animal study reports impressive results in clearing aberrant tau inside neurons and rejuvenating cognitive function, opening up an important new front in damage-repair strategies for maintaining the aging brain.
October 13, 2024
A recent study claimed to find that metformin rejuvenated cognitive function in aging monkeys and lowered biological age on a nonhuman primate biological age clock. The details make the result unconvincing.
October 01, 2024
A supporter asks: Everyone knows that mitochondrial free radicals are a key driver of aging, and antioxidants don’t seem to offer any protection. Birds are supposed to have very clean-burning mitochondria, so should you maybe try to cut them off at the source by re-engineering our mitochondria to be more like those of birds?
August 31, 2024
A supporter asks if “backing up” copies of the mitochondrially-encoded genes in the nucleus is really viable, granted free radical damage in the nucleus. We emphasize the many additional ways that the nuclear copies will be safer than the mitochondrial originals, that the “backup copies” can be backed up again, and how they and additional strategies will buy us time for even better solutions.
August 10, 2024
Aging muscles lose strength above and beyond what would be expected from the mere loss of muscle mass. Accordingly, many drugs have been shown to stimulate muscle growth in older people, but the increased muscle mass consistently fails to translate into increased strength and physical function. To let people live independent lives for longer, we need damage-repair longevity therapeutics to repair the cellular and molecular damage that makes aging muscle dysfunctional.
July 06, 2024
Several pharma companies are currently running clinical trials on damage-repair therapies targeting damaged forms of the protein tau to combat Alzheimer’s disease. But these AmyloSENS therapies only reach tau in the fluid outside of neurons, when what we need is to clear damaged tau inside of them. Fortunately, researchers are beginning to use mRNA — the same revolutionary biotechnology platform of the best COVID vaccines — to develop new LysoSENS therapies to do just that.