Two Polyunsaturated Lipids Demonstrate Senolytic Activity
- They encourage an iron-dependent form of programmed cell death in senescent cells.

- Senescent cells are more prone to oxidation caused by the presence of ferrous ions.
- These two fatty acids encourage a metabolic pathway that hastens this process and damages these cells’ membranes, leading to death by ferroptosis.
- This approach appears to have less systemic toxicity than other senolytics.
A new study identified two polyunsaturated fatty acids, α-eleostearic acid (α-ESA) and α-ESA methyl ester (α-ESA-me), that showed senolytic activity in cell cultures and a mouse model [1].
An anti-aging strategy
Cellular senescence is one of the most critical aging-related processes. Senescent cells, which accumulate with age, are arrested in the cell cycle and are resistant to cell death. The inflammatory senescence-associated secretory phenotype (SASP) factors they release have detrimental effects on the surrounding cellular environment and contribute to many age-related diseases, such as diabetes, cancer, osteoarthritis, and Alzheimer’s disease [2, 3, 4].
Targeting senescent cells has been widely investigated as an anti-aging strategy. While progress has been made and several senolytics that eliminate senescent cells have been identified, many have substantial side effects, preventing their widespread use [5, 6, 7]. Therefore, there is still a need to identify safer and effective options. The researchers of this study focused on fatty acids, natural compounds with various therapeutic effects [8], and investigated their potential as senolytics.
In search of senolytics
The researchers began their study by using senescent cell cultures to screen fatty acids that were previously reported to have health benefits and dietary functions [8]. They observed that the structural features of certain fatty acids were correlated with their potential for senolytic activity.
While there doesn’t seem to be a simple correlation between senolytic activity and carbon chain length, other structural features, such as the position and configuration of double bonds in fatty acids, appeared to influence senolytic activity, with some fatty acids showing senolytic activity in one configuration but not the other. Esterification and conjugation, in which double bonds are not separated by multiple single bonds but occur in an alternating pattern, also affected senolytic activity; for example, most unconjugated fatty acids lacked senolytic activity.
Based on this screen, two fatty acids were selected: the most potent senolytic, α-eleostearic acid (α-ESA), and the most selective senolytic, α-ESA methyl ester (α-ESA-me), both of which are 18-carbon conjugated fatty acids.
Senolysis without toxicity
Identified fatty acids were further tested in various models, including mice and human cell lines with senescence induced by multiple stressors. Those tests confirmed robust senolytic activity of α-ESA and α-ESA-me; however, there was some cell-specific variability. There were also some differences between the two fatty acids. For example, α-ESA showed greater potency and induced senolysis more rapidly, whereas α-ESA-me showed higher selectivity and a more stable, long-lasting effect.
In addition to cell culture testing, the researchers tested the two fatty acids in naturally aged mice (20-22 months) by treating them for 5 days. α-ESA-me was more effective in reducing tissue senescence, especially in the liver and heart. In even older (32-month) mice, “α-ESA-me significantly reduced senescence and SASP factors in multiple tissues” with the strongest effects in kidney, liver, and lung tissues.
Since the effect of α-ESA-me was stronger, it was further tested in progeric mice. A short-term (3-day treatment) led to a reduction in senescence markers and SASP factors. α-ESA-me long-term treatment (three times per week for 6 weeks, starting at 10 weeks of age) led to decreased DNA damage, senescence, and SASP markers, a reduction in the composite score of aging symptoms, and an increase in the number of proliferating cells, all without showing systemic toxic effects. These observations point to α-ESA-me’s senolytic activity and rejuvenating potential.
Going after the mechanism
Furthermore, the researchers investigated the mechanism underlying α-ESA- and α-ESA-me-induced senescent cell death. First, the researchers ruled out the possibility that cells may be converting α-ESA and α-ESA-me into metabolites that play a role in these compounds’ senolytic activity.
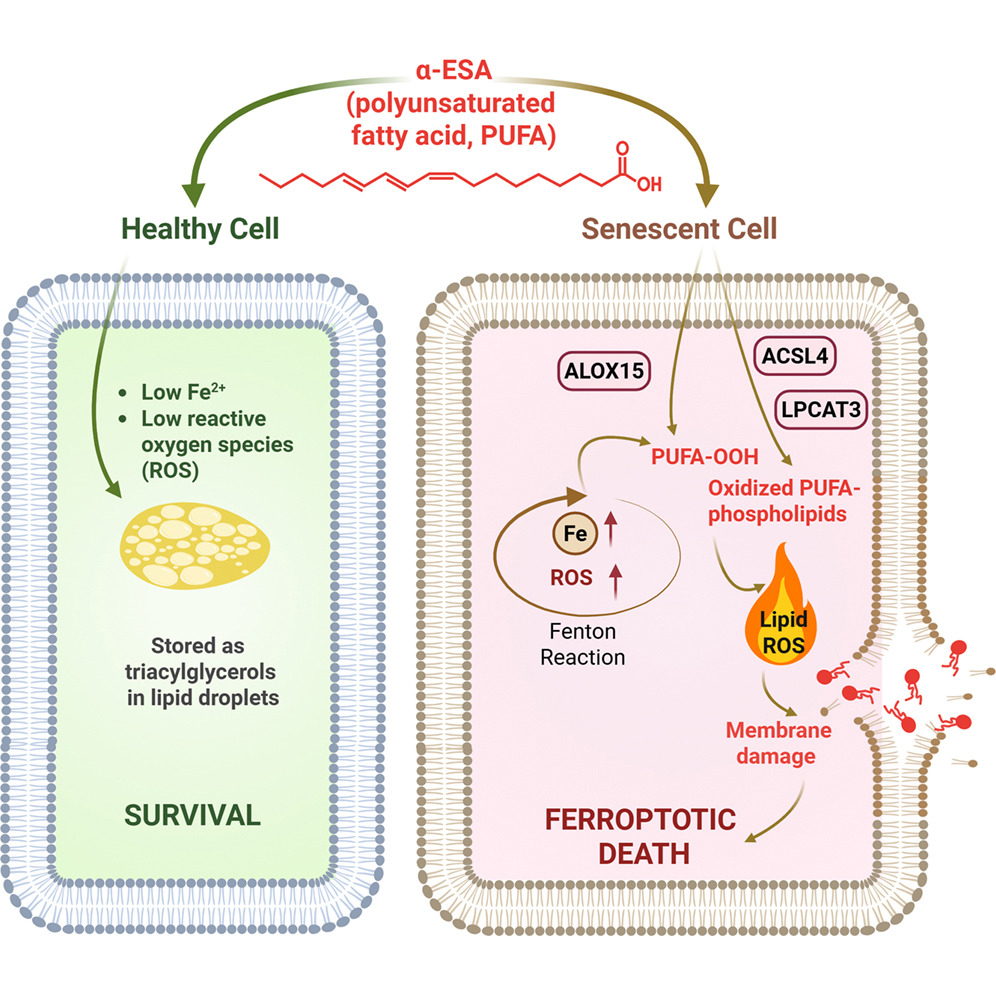
The next line of investigation was whether α-ESA and α-ESA-me act by inducing programmed cell death (apoptosis), as is the case with most senolytics. Investigations into different forms of cell death pointed that it wasn’t apoptosis, but ferroptosis, an “iron-dependent form of programmed cell death triggered by the accumulation of ROS and lipid peroxidation” [9], since blocking ferroptosis-related pathways stopped α-ESAs from inducing cell death. Additionally, a gene expression analysis in the treated senescent cells suggested that α-ESA and α-ESA-me can initiate a ferroptosis-associated transcriptional program and confirmed ferroptosis as a key player in the selective elimination of senescent cells treated with these fatty acids.
To gain further insights, the researchers used a machine learning approach that suggested that these two α-ESAs had molecular pathway interaction profiles similar to those of several known senolytic compounds, such as dasatinib and quercetin, and ferroptosis inducers, such as erastin and sulfasalazine, at a global level. However, compared to those compounds, α-ESAs had a low probability of systemic toxicity while having high oral bioavailability and blood-brain barrier permeability. The results also suggested that α-ESAs might not directly interact with proteins involved in cell-cycle arrest and senescence signaling but may exert an indirect impact.
An in silico analysis identified a potential role for ferroptosis-related pathways and a few proteins (ACSL4, LPCAT3, and ALOX15) as essential for α-ESA-induced senolysis. Inhibiting each of those enzymes significantly protected cells from α-ESA-induced ferroptosis, experimentally confirming that each of those enzymes is necessary for α-ESA’s senolytic effect.
Those results, combined with additional lipidomics experiments, suggested a possible mechanism for α-ESA and α-ESA-me senolytic activity. It appeared that senescent cells have increased levels of ferrous ions and ROS compared to non-senescent cells, and increased iron levels in senescent cells facilitate increased ROS production.
This process is enhanced by α-ESA and α-ESA-me, as their chemical structures make them highly prone to radical formation and propagation, thereby contributing to lipid radical production. In this process, α-ESAs are used as highly oxidizable ‘‘fuel’’ that is further used by the ACSL4-LPCAT3-ALOX15 axis and incorporated in the membrane, leading to the loss of membrane integrity and ferroptotic senolysis.
“This paper is the first to show that lipids can function as senolytics by triggering a distinct form of cell death, called ferroptosis, unlike most current senolytic strategies,” said Paul Robbins, corresponding author of the study, Ph.D., professor at the University of Minnesota Medical School and College of Biological Sciences, and associate director of the Masonic Institute on the Biology of Aging and Metabolism. “Our study reveals that ferroptosis represents a distinct and targetable vulnerability in certain types of senescent cells. Thus, this work opens a new direction for designing senolytic therapies that leverage ferroptotic biology and demonstrates the therapeutic potential of specific bioactive fatty acids.”
Literature
[1] Zhang, L. J., Salekeen, R., Soto-Palma, C., Elsallabi, O., Ye, H., Hughes, B., Zhang, B., Nunes, A., Lee, K.-A., Xu, W., Mohamed, A., Piepgras, E., McGowan, S. J., Angelini, L., O’Kelly, R., Han, X., Niedernhofer, L. J., & Robbins, P. D. (2026). Polyunsaturated lipid senolytics exploit a ferroptotic vulnerability in senescent cells. Cell Press Blue, 100004.
[2] Childs, B. G., Durik, M., Baker, D. J., & van Deursen, J. M. (2015). Cellular senescence in aging and age-related disease: from mechanisms to therapy. Nature medicine, 21(12), 1424–1435.
[3] Baker, D. J., Wijshake, T., Tchkonia, T., LeBrasseur, N. K., Childs, B. G., van de Sluis, B., Kirkland, J. L., & van Deursen, J. M. (2011). Clearance of p16Ink4a-positive senescent cells delays ageing-associated disorders. Nature, 479(7372), 232–236.
[4] He, S., & Sharpless, N. E. (2017). Senescence in Health and Disease. Cell, 169(6), 1000–1011.
[5] Prašnikar, E., Borišek, J., & Perdih, A. (2021). Senescent cells as promising targets to tackle age-related diseases. Ageing research reviews, 66, 101251.
[6] Zhang, L., Pitcher, L. E., Prahalad, V., Niedernhofer, L. J., & Robbins, P. D. (2021). Recent advances in the discovery of senolytics. Mechanisms of ageing and development, 200, 111587.
[7] Kirkland, J. L., & Tchkonia, T. (2020). Senolytic drugs: from discovery to translation. Journal of internal medicine, 288(5), 518–536.
[8] Kremmyda, L. S., Tvrzicka, E., Stankova, B., & Zak, A. (2011). Fatty acids as biocompounds: their role in human metabolism, health and disease: a review. part 2: fatty acid physiological roles and applications in human health and disease. Biomedical papers of the Medical Faculty of the University Palacky, Olomouc, Czechoslovakia, 155(3), 195–218.
[9] Dixon, S. J., Lemberg, K. M., Lamprecht, M. R., Skouta, R., Zaitsev, E. M., Gleason, C. E., Patel, D. N., Bauer, A. J., Cantley, A. M., Yang, W. S., Morrison, B., 3rd, & Stockwell, B. R. (2012). Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell, 149(5), 1060–1072.