Why Fast-Cycling Skin Cells Decrease With Age
- The extracellular matrix does more than hold skin together.
- The protein fibulin-5 is crucial to invoking the YAP pathway and maintaining the populations of fast-cycling stem cells, which naturally decrease with age.
- Mice without this protein have skin that ages significantly faster than that of wild-type mice.
In Aging Cell, researchers have described how one dermal protein is related to maintaining the populations of fast-cycling skin cells and preserving skin integrity.
Not all skin cells are the same
The outer layer of the skin (the epidermis) is naturally regenerated by two distinct populations of stem cells: slow-cycling and fast-cycling [1]. The former are distinguished by the expression of DLX1, while the latter express SLC1A3 [2]. However, with aging, the fast-cycling type begins to decline [3].
This decline occurs alongside several other markers of skin aging. The extracellular matrix (ECM), most known for maintaining tissue integrity and well-known to be damaged with aging, also has significant effects on the behavior of skin cells [4]. Integrins, which give signals to cells about the extracellular environment, decline with aging as well [5]. Further work has found that one particular extracellular matrix component, fibulin-7, is crucial to allowing the proliferation of fast-cycling skin cells [3].
This work focuses on a related protein, fibulin-5, which was also found to decrease with aging [6] and has been documented to directly interact with integrins [7]. This paper, then, set out to demonstrate that it may also be related to the loss of fast-cycling cells.
Mice without fibulin-5 age faster
The researchers created a strain of mice that do not express fibulin-5. Even when young, these mice had loose skin much like that of much older wild-type mice. Additionally, their coats were browner from their usual black, their hair was thinner, and they had less body weight. By 12 months of age, their skin was significantly thinner than that of wild-type mice.
At very young ages, these mice had similar proportions of interscale and scale areas that represent the homes of slow-cycling and fast-cycling skin cells. However, at 12 months, the scale areas were significantly diminished compared to those of wild-type mice, representing a decline in fast-cycling cells.
The modified mice also had significantly diminished amounts of many genes needed for proper skin maintenance, including genes related to replication, cellular adhesion, formation of the extracellular matrix, and one form of signaling. Inflammatory cytokines and a different signaling pathway were upregulated instead. These changes were found to have many aspects in common with ordinary aging.
There was also a significant decrease in the integrity of the ECM at the junction between the epidermal and dermal layers of skin. Like in naturally aged mice, two related integrins and a crucial collagen were downregulated; however, unlike the naturally aged mice, the mice without fibrulin-5 had another integrin downregulated as well. The researchers found that the interaction between integrin β3 and the cellular adhesion molecule nectin-3 was diminished in both naturally aged wild-type mice and in the fibrulin-5 knockout mice.
Cells need to YAP at each other
The overall loss of fast-cycling skin cells was found to be related to the YAP intercellular signaling pathway. There was a decrease in YAP in the mice deficient in fibrulin-5, aged wild-type mice, and skin cells derived from human patients. Using verteporfin to inhibit YAP in wild-type mice significantly decreased their populations of fast-cycling skin cells, and using verteporfin on human skin cells decreased their expression of SLC1A3 as well. Directly exposing these cells to fibulin-5, on the other hand, promoted the expression of SLC1A3, signifying that this compound promotes a fast-cycling state.
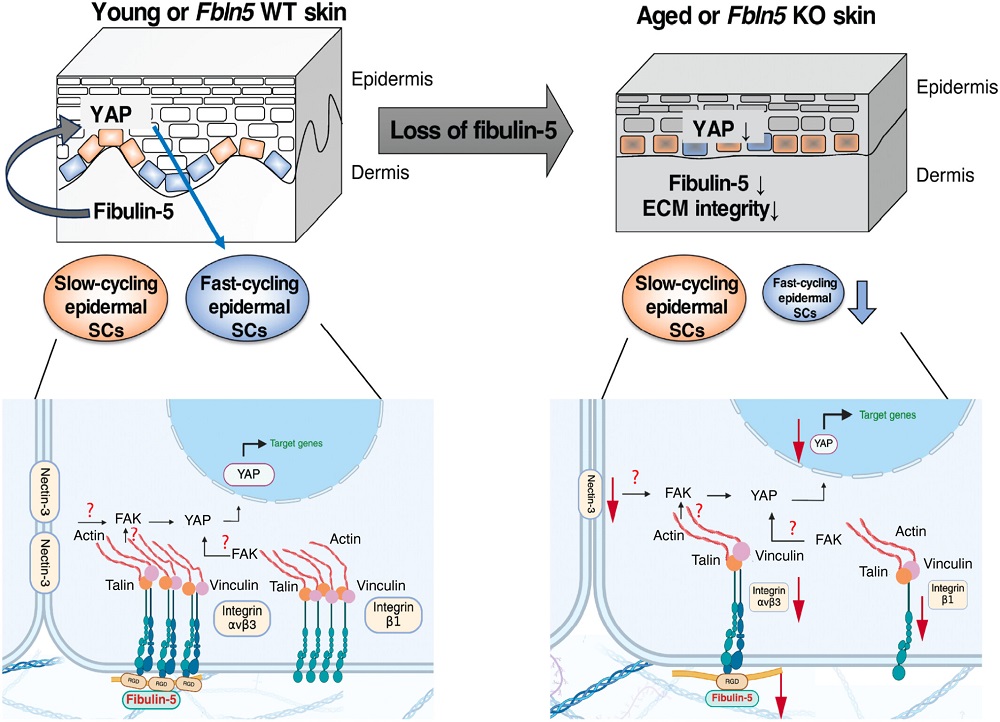
The researchers, however, did not administer fibulin-5 to living animals, nor did they perform any experiments that increase YAP in mice. Such future work will determine if this is a potential path towards rebalancing these cellular populations and potentially treating this form of skin aging in people.
Literature
[1] Ghuwalewala, S., Lee, S. A., Jiang, K., Baidya, J., Chovatiya, G., Kaur, P., … & Tumbar, T. (2022). Binary organization of epidermal basal domains highlights robustness to environmental exposure. The EMBO journal, 41(18), EMBJ2021110488.
[2] Sada, A., Jacob, F., Leung, E., Wang, S., White, B. S., Shalloway, D., & Tumbar, T. (2016). Defining the cellular lineage hierarchy in the interfollicular epidermis of adult skin. Nature cell biology, 18(6), 619-631.
[3] Raja, E., Changarathil, G., Oinam, L., Tsunezumi, J., Ngo, Y. X., Ishii, R., … & Sada, A. (2022). The extracellular matrix fibulin 7 maintains epidermal stem cell heterogeneity during skin aging. The EMBO Reports, 23(12), EMBR202255478.
[4] Wang, Y., Kitahata, H., Kosumi, H., Watanabe, M., Fujimura, Y., Takashima, S., … & Natsuga, K. (2022). Collagen XVII deficiency alters epidermal patterning. Laboratory investigation, 102(6), 581-588.
[5] Giangreco, A., Goldie, S. J., Failla, V., Saintigny, G., & Watt, F. M. (2010). Human skin aging is associated with reduced expression of the stem cell markers β1 integrin and MCSP. Journal of Investigative Dermatology, 130(2), 604-608.
[6] Kadoya, K., Sasaki, T., Kostka, G., Timpl, R., Matsuzaki, K., Kumagai, N., … & Amano, S. (2005). Fibulin‐5 deposition in human skin: decrease with ageing and ultraviolet B exposure and increase in solar elastosis. British Journal of Dermatology, 153(3), 607-612.
[7] Budatha, M., Roshanravan, S., Zheng, Q., Weislander, C., Chapman, S. L., Davis, E. C., … & Yanagisawa, H. (2011). Extracellular matrix proteases contribute to progression of pelvic organ prolapse in mice and humans. The Journal of clinical investigation, 121(5), 2048-2059.