Rejuvenation Roundup November 2023
As researchers and biotech companies continue to work on the therapies and technologies that may let people live longer lives, much other research tells us about the lifestyle habits that people are doing to lengthen them – or shorten them – in the meantime. Here’s what we’ve learned in November.
LEAF News
 An Anonymous Longevity Enthusiast Unmasks Himself: A prolific anonymous poster who became popular in the longevity community has revealed his identity, and other longevity luminaries have chimed in. Anonymity is seldom practiced in the longevity community.
An Anonymous Longevity Enthusiast Unmasks Himself: A prolific anonymous poster who became popular in the longevity community has revealed his identity, and other longevity luminaries have chimed in. Anonymity is seldom practiced in the longevity community.
Research Roundup
Bacteria from Alzheimer’s Patients Induced Symptoms in Rats: New research published in Brain investigated the influence of fecal microbiota transplants from Alzheimer’s patients on cognitive function and neurogenesis in rats and human cell cultures.
 Hyperbaric Oxygen Therapy Improves Bone Health in Rats: Scientists have demonstrated that hyperbaric oxygen therapy can reverse some aspects of bone deterioration caused by aging and obesity in rats. This involves placing the patient in a chamber with elevated levels of oxygen and atmospheric pressure.
Hyperbaric Oxygen Therapy Improves Bone Health in Rats: Scientists have demonstrated that hyperbaric oxygen therapy can reverse some aspects of bone deterioration caused by aging and obesity in rats. This involves placing the patient in a chamber with elevated levels of oxygen and atmospheric pressure.
Predicting Future Frailty with an Epigenetic Clock: An accepted manuscript in the Journals of Gerontology has described how a third-generation epigenetic clock is a predictor of future frailty.
 Alcohol Consumption Increases Epigenetic Age Acceleration: A new study has found that higher levels of long-term alcohol consumption are associated with a larger gap between the person’s biological and chronological age, but not in the young.
Alcohol Consumption Increases Epigenetic Age Acceleration: A new study has found that higher levels of long-term alcohol consumption are associated with a larger gap between the person’s biological and chronological age, but not in the young.
Tweaking the Growth of Stem Cells for Better Therapies: Researchers publishing in ACS Nano have described how culturing stem cells on nanogratings instead of flat substrates changes the effects of the extracellular vesicles (EVs) they send, potentially paving the way to a new system of therapies.
 Lifestyle Interventions Against Cellular Senescence: A new review summarizes our knowledge about lifestyle interventions, such as exercise, healthy diet, and good sleep, that can possibly reduce cellular senescence.
Lifestyle Interventions Against Cellular Senescence: A new review summarizes our knowledge about lifestyle interventions, such as exercise, healthy diet, and good sleep, that can possibly reduce cellular senescence.
Olive Oil Fights Cognitive Decline in Clinical Trials: In a recent review published in Frontiers in Nutrition, the researchers reviewed studies linking olive oil consumption to cognitive performance.
 Cells Regularly Donate Mitochondria to One Another: In a review published in Nature, researchers have explained how and why cells transfer mitochondria to one another, proposing that this transfer allows organisms to be more resilient against energy deficiencies.
Cells Regularly Donate Mitochondria to One Another: In a review published in Nature, researchers have explained how and why cells transfer mitochondria to one another, proposing that this transfer allows organisms to be more resilient against energy deficiencies.
Mediterranean, Keto, and Plant-Based Diets Vs. Cancer: A new review summarizes what we know about the Mediterranean, keto, and plant-based diets and their effects on cancer risk and progression.
 Targeting Metabolism to Protect Against Stroke: Japanese researchers publishing in Aging have described how improving the AMPK metabolic pathway improves outcomes in a rat model of stroke.
Targeting Metabolism to Protect Against Stroke: Japanese researchers publishing in Aging have described how improving the AMPK metabolic pathway improves outcomes in a rat model of stroke.
A Phytomolecule Improves Tendon Regeneration in Rats: A new study has identified a molecule found in Saposhnikovia root as a potent mediator of tendon regeneration in rats. The results were also replicated in human cells.
 Promoting Health and Longevity Through Diet: A review published in the Journal of Internal Medicine summarized current knowledge on the impact of dietary factors on chronic diseases and longevity.
Promoting Health and Longevity Through Diet: A review published in the Journal of Internal Medicine summarized current knowledge on the impact of dietary factors on chronic diseases and longevity.
Senolytics May Work Against Long COVID in the Brain: A new publication in Nature Aging has used organoids and model mice to discover that senolytics may be effective treatments for neurodegeneration caused by the post-infection syndrome known as long COVID.
 Plant-Based Alternatives Are Associated With Less Disease: A new meta-analysis has found that in many cases, switching from an animal product to a plant-based alternative is associated with less cardiovascular, diabetes, and all-cause mortality risk
Plant-Based Alternatives Are Associated With Less Disease: A new meta-analysis has found that in many cases, switching from an animal product to a plant-based alternative is associated with less cardiovascular, diabetes, and all-cause mortality risk
Pushing Forward on Understanding Cellular Decline: Researchers publishing in Nature Aging have investigated one of the core biological reasons behind the decline of stem cells’ ability to proliferate.
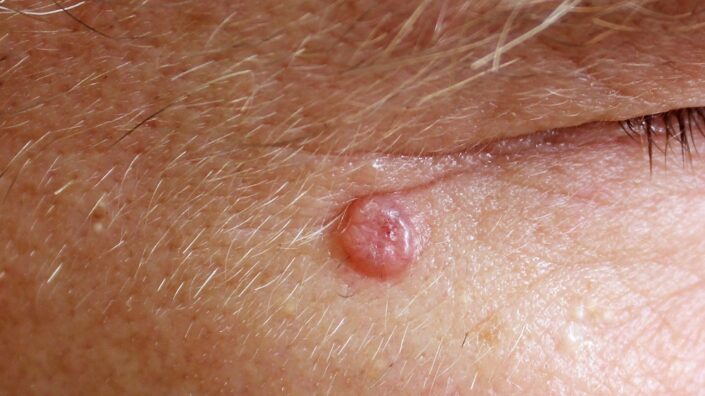
 A Lack of Skin Collagen May Encourage Cancer Growth: A paper published in Nature has described how the aging extracellular matrix causes physical changs to the skin that make it easier for cancer to grow.
A Lack of Skin Collagen May Encourage Cancer Growth: A paper published in Nature has described how the aging extracellular matrix causes physical changs to the skin that make it easier for cancer to grow.
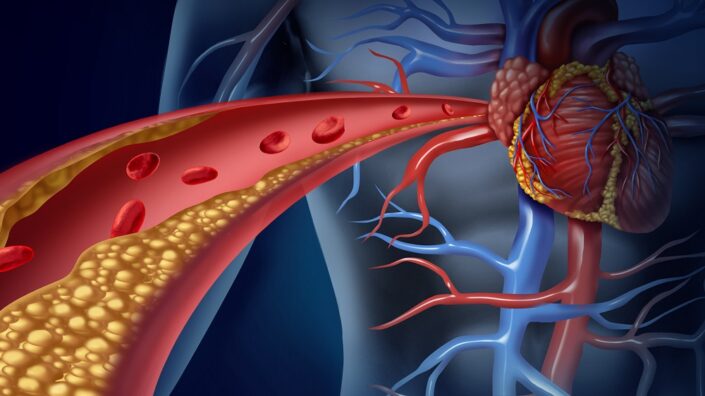
Young People More Vulnerable to Atherosclerosis Risk Factors: One study suggests that increases in LDL cholesterol and systolic blood pressure elevate the risk of atherosclerosis progression more in younger than in older people. The good news is that this can be reversed.
 Dual-Purpose Molecular Targets for Cancer and Aging: A study published in Aging Cell applied an artificial intelligence target discovery platform to aid in identifying potential dual-purpose targets for anti-aging and anti-cancer treatments.
Dual-Purpose Molecular Targets for Cancer and Aging: A study published in Aging Cell applied an artificial intelligence target discovery platform to aid in identifying potential dual-purpose targets for anti-aging and anti-cancer treatments.
Some HIV-Fighting Drugs May Also Fight Aging: Publishing in Ageing Research Reviews, a team of researchers including Vera Gorbunova has described how a class of drugs originally used to treat HIV might be used to affect some of the hallmarks of aging.
 Aging Might Blunt the Effects of Fasting: Scientists have found that in killifish, old age is associated with a dampened response to fasting and identified the protein that might be responsible. A similar mechanism might exist in humans.
Aging Might Blunt the Effects of Fasting: Scientists have found that in killifish, old age is associated with a dampened response to fasting and identified the protein that might be responsible. A similar mechanism might exist in humans.
Predicting a Species’ Longevity Through Epigenetics: A preprint study published on bioRxiv discusses how a species’ maximum lifespan can be predicted by its epigenetics, showing that these differences are largely unaffected by interventions.
 Mammals Accumulate Epigenetic Noise With Age: In Nature Communications, researchers have explained how epigenetic drift and disorder are associated with shorter lifespans across species and that some species accumulate this noise considerably faster than others.
Mammals Accumulate Epigenetic Noise With Age: In Nature Communications, researchers have explained how epigenetic drift and disorder are associated with shorter lifespans across species and that some species accumulate this noise considerably faster than others.
Isoleucine Restriction Boosts Lifespan in Mice: A new study reports a massive 33% increase in median lifespan in male genetically heterogeneous mice following prolonged isoleucine restriction.
 Music Therapy for Treating Alzheimer’s Disease: The authors of a new review have analyzed randomized controlled trials that evaluated the impact of music therapy on the cognitive functions of people with Alzheimer’s.
Music Therapy for Treating Alzheimer’s Disease: The authors of a new review have analyzed randomized controlled trials that evaluated the impact of music therapy on the cognitive functions of people with Alzheimer’s.
Running Is Associated With Better Health Biomarkers: In a recent publication in PLOS One, researchers have described the metabolic effects on running in generally healthy people. A lack of activity is a known risk factor for early mortality.
Implanting mechanically reprogrammed fibroblasts for aged tissue regeneration and wound healing: Transcriptome analysis, combined with chromatin biomarkers, revealed that implanted cells upregulated tissue regeneration and wound healing pathways. These results provide a novel, nongenetic, partial reprogramming of aged cells for cell-based therapies in regenerative medicine.
Sleep regularity and mortality: a prospective analysis in the UK Biobank: Irregular sleep-wake patterns are associated with higher mortality risk, including mortality due to cancer and cardiovascular disease.
Calorie restriction reduces biomarkers of cellular senescence in humans: These results advance the understanding of the effects of CR in humans and further support a link between cellular senescence and metabolic health.
Mechanisms underlying retardation of aging by dietary energy restriction: Overnutrition disrupts FoxO- and Npy-associated metabolic and mitochondrial bioenergetic adaptive processes, causing the acceleration of aging and related diseases.
Effect of Dietary Sodium on Blood Pressure: This crossover trial assesses whether high-sodium and low-sodium dietary interventions affect 24-hour ambulatory blood pressure measures among middle-aged to elderly adults with normotension, controlled hypertension, uncontrolled hypertension, or untreated hypertension.
Dietary Sodium Intake and Risk of Incident Type 2 Diabetes: To fill the knowledge gap of the relation between long-term dietary sodium intake and type 2 diabetes (T2D), the researchers evaluated the association between the frequency of adding salt to foods, a surrogate marker for evaluating the long-term sodium intake, and incident T2D risk.
Effects of exercise interventions on cognitive functions in healthy populations: A systematic review and meta-analysis: Chronic exercise intervention is a non-pharmacological therapy suggested to improve cognitive function in various populations.
Efficacy of morning versus afternoon aerobic exercise training on reducing metabolic syndrome components: High-intensity aerobic exercise training in the morning in comparison to training in the afternoon is somewhat more efficient at reducing cardiometabolic risk factors (i.e. systolic blood pressure and insulin sensitivity).
Dose-response of accelerometer-measured physical activity, step count, and cancer risk in the UK Biobank: a prospective cohort analysis: In this sample from the UK Biobank, higher total physical activity and daily step count were associated with lower risk of physical-activity-related cancers. Findings suggest additional physical activity time, irrespective of intensity, may be beneficial. Increasing low intensity activity time and increasing daily step counts could be practical.
Effect of physical activity on risk of Alzheimer’s disease: A systematic review and meta-analysis of 29 prospective cohort studies: PA intervention reduces the incidence of AD, but merely in moderate to vigorous PA with follow-up of less than 15 years, thus conditionally recommending the popularization of PA as a modifiable lifestyle factor to prevent AD.
Tumor cell-induced macrophage senescence plays a pivotal role in tumor initiation followed by stable growth in immunocompetent condition: This paper reports that tumor growth can be inhibited by externally administered NMN against a macrophage senescence-like state that occurs in the very early stages of tumor-initiating cell development.
Fermented cereal-origin gerobiotic cocktails promote healthy longevity in Caenorhabditis elegans: Several single-strain gerobiotics have proven to be beneficial in alleviating aging and age-related functional declines across species, but multistrain/multispecies gerobiotics appear to be even more useful.
Antibiotics that target mitochondria extend lifespan in C. elegans: Mitochondrial inhibitors (i.e., mitochondria-targeting antibiotics) could abrogate aging and extend lifespan in C. elegans.
A Combination Study of Pre- and Clinical Trial: Seaweed Consumption Reduces Aging-Associated Muscle Loss: This treatment helps maintain muscle mass and delays muscle wasting during aging, suggesting it as a potent nutritional strategy to protect against aging-associated sarcopenia.
Microvesicles-hydrogel breaks the cycle of cellular senescence by improving mitochondrial function to treat osteoarthritis: Through responding to ROS levels, this hydrogel intelligently releases MVs and enhances mitochondrial function in chondrocytes to improving cellular senescence.
In vivo reprogramming leads to premature death linked to hepatic and intestinal failure: In transgenic mice, continuous induction of the reprogramming factors in vivo leads to hepatic and intestinal dysfunction, resulting in decreased body weight and contributing to death within 1?week.
Mid-old cells are a potential target for anti-aging interventions in the elderly: In this study, the authors introduce the concept of a unique cellular subtype within the organic stroma, which does not conform to a typical young or senescent cell.
Electric currents of 448?kHz upregulate anti-senescence pathways in human dermal fibroblasts: CRET treatment improves a number of functions related to migration and proliferation, and it reduces age-related cellular changes in human dermal fibroblasts.
The longevity bottleneck hypothesis: Could dinosaurs have shaped ageing in present-day mammals?: Although mammals, such as humans, can evolve long lifespans, they do so under constraints dating to the dinosaur era.
Survival and longevity of European rulers: geographical influences and exploring potential factors, including the Mediterranean diet: Although the Mediterranean diet is often associated with health benefits, these findings indicate that it alone may not account for differences in ruler longevity across regions.
News Nuggets
 Rejuve’s Longevity App Welcomes Peptide Bioregulator: ‘Longevity’, an app designed to empower everyone to live longer, healthier lives, welcomes its latest partner, Peptide Bioregulator, a high-quality supplement company.
Rejuve’s Longevity App Welcomes Peptide Bioregulator: ‘Longevity’, an app designed to empower everyone to live longer, healthier lives, welcomes its latest partner, Peptide Bioregulator, a high-quality supplement company.