Everlasting boredom is a common concern when it comes to the idea of extended human lifespans. There are many people who expect life to become empty and meaningless with time because everything has been already experienced; there is nothing else to discover, nothing else can surprise or please.
This idea has become so ingrained in our collective psyche that its mark appears constantly in our most popular works of fiction. The elves from the Lord of the Rings, for example, are described as lacking passion and ambition because of their extended lives, as compared to humans whose short lifespans impel them to bold action. Another omnipresent trope is the tale of the tortured immortal who would do anything to become human again or to end the curse with the finality of death.
The thinking goes that people living longer than a century will become tired of such hollow lives, eventually leading to the decision to terminate their pitiful existences. The loss of the thirst for life in the elderly is supposed to illustrate the probability of this outcome.
So, let’s consider these concerns and see if there is any scientific basis to the idea that increased lifespans would lead to a boring existence of never-ending tedium.
Will a longer life mean that I am more bored than I am now?
The ‘hedonic treadmill’ probably isn’t a title that grabs your attention. Despite its name, it isn’t a type of naked exercise routine, but it is an important factor in why you will not get more bored just because you live longer.
At its heart, the hedonic treadmill suggests that our base level of happiness will always remain the same, despite perceived positive changes. If something ‘good’ happens we will eventually return to that same base level of happiness and we will seek the next ‘good’ thing. An example of this is salary; we think we will be happier if we make more money, but when we do make more money we start to seek the next money milestone. The same thing applies to boredom where we will always be seeking the next buzz but our base level of boredom will never change and we will never be more bored just because we live longer.
With the rapid march of technology, there has been and will continue to be a nearly endless number of new things to experience and enjoy. Our society and technology rarely remain still for long so there will always be plenty to see, experience and do just as this is now. In fact, when you consider it, the pace of progress and change is increasing.
Consider just how much progress has been made since the previous century, we went from the first powered flight to the jet engine in forty years which led to a boom in tourism in the 1950s and accessible holidays to exciting new places for many people. These people would have rarely left their hometowns before this and now they could explore far-flung places and experience things they never dreamed of thanks to progress and technology.
Computers have opened up a world of on-demand entertainment, from video games, streamed films and television, social media and more. The sum of human knowledge is now at our fingertips thanks to the internet and we can learn about a vast range of subjects and enjoy almost unlimited entertainment. The types of entertainment available via computers is also growing with virtual reality and augmented reality become increasingly sophisticated and offering another vista of experiences to enjoy.
The world will not stop changing and there will be a near unlimited range of things to see and do, so no you will not be more bored than you are now no matter how long you live. Importantly, boredom is natures way of telling you there is a problem, we will discuss why boredom happens in the following sections and how to recognize and tackle it.
What is boredom, and why do we experience it?
First of all, let’s remember that all the feelings we have were developed during our evolution as a species to serve adaptation. Emotions represent the conclusion of a very fast overall analysis of a situation, guiding our responses and helping to choose a proper reaction.
Anger, for instance, is the reflexive response to meeting an obstacle, threat, or tense social situation and is designed to temporarily increase an organism’s strength [1] and resistance to injury in case of a conflict – this is why anger raises a person’s heart rate and blood pressure [2], while blood parameters change to prevent excessive bleeding in case of an injury [3].
Boredom, too, serves an evolutionary purpose. Feeling bored means that an individual’s needs for stimuli are not being fulfilled; new experiences are not being explored because, for some reason, it is difficult to engage in a new activity [4]. It has been hypothesized that boredom was developed to regulate goal-pursuit by temporarily boosting mindfulness and increasing one’s sensitivity to the value of alternative goals.
There are two main triggers of boredom: a lack of challenge in our current activities (meaningless) and non-structured time (nothing to do).
Lack of a challenge leads to boredom
 Is it possible that, in the future, we might be locked into a completely safe, stable and tedious environment for an indefinitely long time? Could it be that life experiences are finite? Might we really find ourselves in a situation where we will lose the vital needs that push us forward and drive our desire for new experiences and activities today?
Is it possible that, in the future, we might be locked into a completely safe, stable and tedious environment for an indefinitely long time? Could it be that life experiences are finite? Might we really find ourselves in a situation where we will lose the vital needs that push us forward and drive our desire for new experiences and activities today?
It is worth noting that, even if science manages to bring the aging processes under medical control and we can achieve negligible senescence, it still does not imply indestructibility. Even if people do not die from age-related diseases anymore, there will still be dangers, such as accidents, wars, murders, infectious diseases, dangerous animals, and natural disasters, such as hurricanes, earthquakes, and tsunamis.
Even if human civilization develops a sophisticated system to control natural phenomena, there will still be such dangers as asteroid impacts and supernovae. Our own Sun will also eventually heat up, expand many times its current size and engulf the Earth later in its lifetime. If that was not bad enough, our galaxy will also eventually collide with the Andromeda galaxy, which is heading our way!
If we focus only on our immediate surroundings, it may seem that we have nothing to be concerned about, that we are living in safety and the world is friendly and stable. However, the universe is not a safe place. As humans, we have had to invent language, weapons, fire, clothes, houses, electricity, antibiotics, and computers. We have had to protect ourselves against many dangers; we have had to take control of our lives to survive as a species.
As long as there are uncontrollable factors that surpass our adaptations, there will always be a drive for exploration and development. Given all this, it is very unlikely that we are going to be able to control our universe anytime soon – which means that we will have more than enough interesting challenges to keep us busy for many human lifespans.
Does this mean that we will never feel bored at some point in time?
No, it does not. In fact, we will likely feel bored regularly, because this is an evolutionary tool to foster our self-development as individuals. It makes us seek out more meaningful activities, such as leaving a current career for something new or starting a new project or setting a new goal.
It appears that in order to enjoy longer lives, we will first have to master boredom. If we learn how to identify and pursue more fulfilling activities faster, we will spend less time feeling bored. The first step is to admit when you are bored and recognize what it means: “This activity does not let you live to your full potential. Maybe it is time to try doing something more creative and engaging?”
So how do you discover a new vocation to set a higher goal?
There are several strategies, depending on the amount of challenge you need. The easiest way is to engage in a new hobby or seek new opportunities at work. Ask your boss at work to occasionally give you some small tasks beyond your usual duties. This way, you can try doing something new and more challenging while remaining in your comfort zone. If that is not enough, you could look for opportunities for transfer or promotion within your company.
If you wish to contribute more significantly to business and society, it may be useful to get into the habit of reading industry reports and magazines in order to learn about bottlenecks and promising new niches. There is always the need for new products and services that an entrepreneur can bring to market. You don’t have to build your own company right away; it might be more convenient to join an existing startup and unite your efforts with other people who share your new values and goals.
There are many global problems identified by our society which have sets of corresponding goals, such as the ones described in the Sustainable Development Goals Agenda 2016-2030. Solving global problems might offer a very strong feeling of purpose, as these types of activities can affect the lives of millions of people.
There is no need to be concerned with the scale of a task; every good thing we see around us is the result of a collective effort in which everyone has performed to the best of their abilities and knowledge to ensure common success.
Too much challenge causes boredom
Nature seeks to save resources and increase productivity. This is why we have developed mechanisms to limit our activities when the chance of success is perceived as small. Remember the fable about the fox and the grapes? As the grapes are hanging too high to reach, the fox gives up saying that the grapes were not ripe enough anyway.

Our world is complex, and that complexity is increasing. The current information storm is a perfect example. The ancient library of Alexandria is estimated to have contained from 40 to 400 thousand scrolls [5-6]. If we presume that one scroll consists of approximately 10-20 pages, then 10 scrolls would be the equivalent of 1 modern book. We can estimate then that the Alexandria library may have contained an equivalent of 4 to 40 thousand books. A single person could read 4,000 books in a current human lifetime, assuming one book a week for 80 years. In 2010, Google calculated the number of unique books in the world, and there were about 130 million. The amount of information that a person must consume to be able to push scientific and technological progress forward has significantly increased over the last few decades. An approximate calculation shows that to become a Ph.D, a person might need to read about 700 books, which would take about 13 years.
It is, therefore, no surprise that many people perceive the world of tomorrow as being too challenging – and the lack of enthusiasm to live for a hundred years or longer is a sort of a defensive reaction to this excessive challenge. However, let’s remember that newly identified needs represent an excellent opportunity for entrepreneurs to invent and market necessary products and services. If there is the need for faster and easier consumption of information, the corresponding services will emerge.
What do you do if you lack the self-confidence to pursue your new goal?
The school system was the first social adaptation to the increased need for education. Today, to help people adapt, many countries are adopting the concept of lifelong learning [7]. They are fostering the development of new free education programs (including the programs for senior citizens) and MOOC (Massive Open Online Courses) platforms, such as Coursera, Udemy and Edx.
There are many local MOOC platforms, and the best courses get translated into many languages. These projects allow anyone with a laptop, tablet or smartphone with an Internet connection to acquire new knowledge and skills to enter a new professional field. Furthermore, user activity data is being collected and analyzed in order to constantly improve the learning process and help people get better results in less time.
The promise of intelligence enhancement
Neurophysiologists are studying the processes of thinking and memory and are trying to figure out how to send new data to the brain using a brain-to-brain or brain-to-computer interface. The current level of knowledge enables scientists to make a rat move its tail using input from a human brain [8]. Researchers have also found a way to create false memories in mice [9].
So what about humans? Some medications are known to increase neuron protection and the development of new neural connections, hence reducing the rate of brain deterioration with age or after a trauma and, to some point, increasing learning capacities. These medications are sometimes known as nootropics and include compounds of different origins, from flavonoids found in berries to synthetic drugs. While their efficacy is debated, there is an ongoing effort to produce such performance-enhancing drugs.
There are currently numerous attempts to create non-invasive methods of cognition improvement, including brain electromagnetic stimulation. The first startups to enhance human learning with implants are also already here. So far, it is hoped that the technology might help people suffering from epilepsy (this illness affects their memory processes), but if the memory-boosting implant shows robust proof-of-concept, the implications for its use could be much wider.
As skills are not very different from other types of information in our brains, it may even be possible to acquire new skills by adding that knowledge to the brain via an interface that bypasses the current learning process. This could mean that learning new skills would be dramatically faster than it is now. Of course, this is pure speculation at this point, but the possibilities of this potential technology are clear.
Usually, it takes an average of 12 to 15 to test and register a new drug, and a medical device often requires less time. Therefore, it is possible that the first devices for assisted learning will arrive long before strategies for negligible senescence are developed and available globally.
Therefore, we may perhaps become better problem solvers before we become young supercentenarians. If this happens, it will be easier to organize our lives and get involved in any project we like, so it is unlikely that we will be suffering from boredom for long periods of time.
Non-structured time
Some people might feel that a long life might be boring because they cannot really imagine the distant future and the challenges we might face. What will the world be like, and what topical issues will happen in the next fifty years?
Well, unless you are a well informed futurologist with an expert forecast in your hands, it is hard to even guess how the world will change in the future. What will your typical working day be like 50 years from now? Can you describe it in detail? What do you think fashionable hobbies or pastimes will be in the future? Can you imagine planning a vacation to Mars today, when Martian cities are still only in the portfolios of architects?
It is hard to imagine and answer these questions, and when many people’s minds meet too much uncertainty, they react to this empty space and non-structured time with boredom and anxiety. This uncertainty can inspire a lack of confidence because we don’t know how to deal with the unknown that considering the distant future inspires.
The reason why many futurists are not as concerned about boredom, and often feel the opposite, is because they are excited about the future and want to live as long as possible to see that future. This is because their picture of the future is well developed, and their place in that future is something they have considered and are eager to experience.
They can easily imagine their place in this amazing world of tomorrow and what will keep them busy in that future. For example, futurist Ray Kurzweil has developed complex mathematical models to predict technological progress, and he regularly publishes forecasts with approximate dates for new technology arriving and how it could change the world. He openly expresses his wish to see the distant future, and he takes measures to maintain his health; his approach is described in the book “Transcend: Nine Steps to Living Well Forever“.
Researchers studying time perspective theory indicate that during the course of a person’s life, that person can be focused on different time perspectives: past, present, or future [10]. A person’s mind filters information depending on which time perspective becomes dominant. The adherence to one or another time perspective is a product of lifelong learning; it can change due to education and training.
It is interesting to note that the future time perspective is known to support a healthy lifestyle [11], helps with adherence to prescribed therapy [12], and can promote psychological well-being. It is found to be more common among entrepreneurs, which is not surprising, as entrepreneurship involves a lot of analysis of market forecasts and detailed business development planning [13].
Thus, drawing a more detailed picture of the future (via reading corresponding materials) is a good way to reduce the amount of time spent feeling bored while increasing one’s chances for a better life.
Tired of living – or tired of being ill and dependent?
When discussing longer lives, some people are concerned that increased lifespans might actually be detrimental to a person. The argument holds that living longer makes a person become tired of life; such people often illustrate this idea with the example of old people. However, that argument does not consider the differences between a chronologically and biologically old person versus a chronologically old but biologically younger person after negligible senescence technologies are applied.
| |
Chronologically and biologically old (80+) |
Chronologically old (80+) but negligibly senescent |
| Health |
Many chronic diseases and partial or full disability |
Good health |
| Appearance |
Typical for an old person |
Young or middle-aged |
| Cognitive capacities |
Defined by health status |
Full capacities |
| Skills and knowledge |
Being gradually reduced accordingly to health status |
Constantly increasing |
| Social/Financial status |
Retired (Dependent) |
Working (Independent) |
| Social inclusion |
Limited (Absent) |
Full inclusion |
| Exposure to ageism and abuse |
Very likely |
Unlikely |
| Life expectancy |
115 years |
Indefinite |
| Mood |
Likely lower due to age-related factors e.g., poor health, abuse, social isolation, dependence |
Not affected by age-related factors, depends mostly on personal achievements |
Fig 1. Aging vs negligible senescence. Life expectancy is based on a publication appearing in the journal Nature[14].
We tend to forget how many things represent a challenge for a biologically old person in our society and how biological age and a remaining short lifespan affect one’s quality of life. It takes a lot of courage to admit how badly old people truly feel in their last years. But the sad truth has to be said, and it has to be heard. While cheering up the elderly by telling them that there is still beauty and meaning in being old, we should not forget that biological aging is a leading cause of disease, suffering, and death.
A fascinating journey to health
For many thousands of years, humanity has tried to find a way to maintain good health throughout life. The earliest myths tell the story of how humans have lost their immortality, expressing our intrinsic wish to live in good health for indefinitely long. There is no doubt that this primary wish drives the development of medicine.
Humanity is quite unanimous in this thirst for long-lasting health. In the 20th century, this idea was introduced into the texts of different international treaties regarding health and well-being. For instance, the Constitution of the World Health Organization (which is the United Nations body responsible for healthcare development) defines health as:
A state of complete physical, mental and social well-being and not merely the absence of disease or infirmity
The objective of the World Health Organization is the attainment by all peoples of the highest possible level of health. However, it is impossible to make people healthy if they are still suffering from age-related diseases. Thus, canceling aging is in complete accordance with the universal strategy of health improvement.
Literature
[1] Cox, D. E., & Harrison, D. W. (2008). Models of anger: contributions from psychophysiology, neuropsychology and the cognitive-behavioral perspective. Brain Structure and Function, 212(5), 371-385.
[2] Fernandez, E. (Ed.). (2013). Treatments for anger in specific populations: Theory, application, and outcome. Oxford University Press.
[3] Mittleman, M. A., & Mostofsky, E. (2011). Physical, Psychological and Chemical Triggers of Acute Cardiovascular Events Preventive Strategies. Circulation, 124(3), 346-354.
[4] van Tilburg, W. A., & Igou, E. R. (2016). Boredom Begs to Differ: Differentiation From Other Negative Emotions. Emotion (Washington, DC).
[5] Murray, S. A. (2013). The library: An illustrated history. Skyhorse Publishing, Inc..
[6] Lyons, M. (2011). Books: a living history (p. 224). J. Paul Getty Museum.
[7] UNESCO Institute for Lifelong Learning (UIL). (2014). Medium-term strategy 2014-2021: laying foundations for equitable lifelong learning for all.
[8] Yoo, S. S., Kim, H., Filandrianos, E., Taghados, S. J., & Park, S. (2013). Non-invasive brain-to-brain interface (BBI): establishing functional links between two brains. PloS one, 8(4), e60410.
[9] de Lavilléon, G., Lacroix, M. M., Rondi-Reig, L., & Benchenane, K. (2015). Explicit memory creation during sleep demonstrates a causal role of place cells in navigation. Nature neuroscience.
[10] Stolarski, M., Fieulaine, N., & van Beek, W. (2015). Time perspective theory; Review, research, and application. Cham: Springer International.
[11] Griva, F., Tseferidi, S. I., & Anagnostopoulos, F. (2015). Time to get healthy: Associations of time perspective with perceived health status and health behaviors. Psychology, health & medicine, 20(1), 25-33.
[12] Sansbury, B., Dasgupta, A., Guthrie, L., & Ward, M. (2014). Time perspective and medication adherence among individuals with hypertension or diabetes mellitus. Patient education and counseling, 95(1), 104-110.
[13] Przepiorka, A. (2016). What makes successful entrepreneurs different in temporal and goal-commitment dimensions?. Time & Society, 25(1), 40-60.
[14] Dong, X., Milholland, B., & Vijg, J. (2016). Evidence for a limit to human lifespan. Nature, 538(7624), 257-259.




 First, there are overwhelmingly more “normal” people than tyrants and dictators. Left uncured, the disease would make the lives of the latter miserable and eventually kill them, but it would do the same to the former, innocent “normal” people, including those who live under the tyrants.
First, there are overwhelmingly more “normal” people than tyrants and dictators. Left uncured, the disease would make the lives of the latter miserable and eventually kill them, but it would do the same to the former, innocent “normal” people, including those who live under the tyrants. Third, the high costs and low benefits—not to mention the high chance of failure—of this “strategy” to get rid of tyrants make it even more unjustifiable when you consider the alternatives. If the goal is ending the oppression on the people perpetrated by a dictator, it seems more logical and effective to orchestrate an attack to arrest or kill them than wait until the disease gets them—especially given how slow the disease is.
Third, the high costs and low benefits—not to mention the high chance of failure—of this “strategy” to get rid of tyrants make it even more unjustifiable when you consider the alternatives. If the goal is ending the oppression on the people perpetrated by a dictator, it seems more logical and effective to orchestrate an attack to arrest or kill them than wait until the disease gets them—especially given how slow the disease is. However, waiting for the tyrant to die of old age is also a ridiculous game of chance, namely the chance that, over such a long time, the tyrant wouldn’t manage to find someone to replace them and carry on the tyranny despite his death.
However, waiting for the tyrant to die of old age is also a ridiculous game of chance, namely the chance that, over such a long time, the tyrant wouldn’t manage to find someone to replace them and carry on the tyranny despite his death.

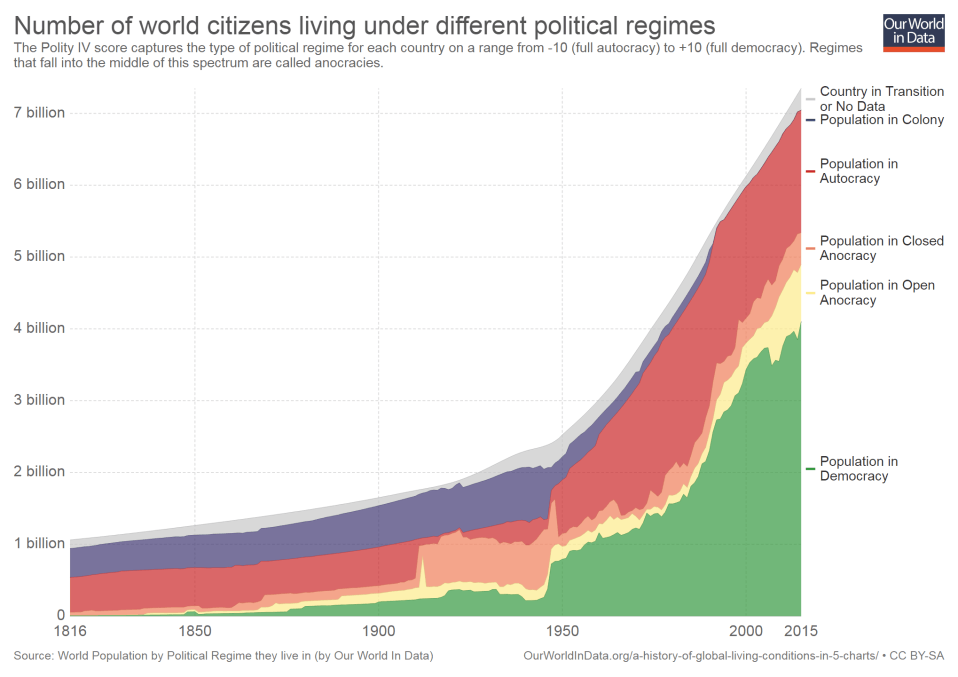





 Is it possible that, in the future, we might be locked into a completely safe, stable and tedious environment for an indefinitely long time? Could it be that life experiences are finite? Might we really find ourselves in a situation where we will lose the vital needs that push us forward and drive our desire for new experiences and activities today?
Is it possible that, in the future, we might be locked into a completely safe, stable and tedious environment for an indefinitely long time? Could it be that life experiences are finite? Might we really find ourselves in a situation where we will lose the vital needs that push us forward and drive our desire for new experiences and activities today?