A Review of How the Heart Ages
- Some parts change completely, while others remain largely the same.
- The cells and morphology of the heart both change with age.
- As muscle cells become senescent, the heart enlarges to compensate for the ineffective tissue.
- Mitochondrial dysfunction decreases available energy and leads to harmful fat accumulation.
- The heart’s natural pacemaker becomes fatty and filled with elastic tissue.
The European Heart Journal has published a review of what happens to the human heart as it ages, noting the cellular effects of mitochondrial dysfunction and cellular senescence along with more visible changes such as hypertrophy and fibrosis.
A constantly working organ
The reviewers begin this paper by noting the constant work of the heart, as it must unceasingly pump blood for us to be able to live. Unfortunately, this organ is well-known to fail with age; while only roughly 1% of people under 55 suffer heart failure, it occurs in over 10% of people over 70 [1].
Understanding and classifying the myriad ways in which the heart ages is a crucial first step in developing therapies against age-related diseases. To that end, members of the International Consortium to Classify Ageing-Related Pathologies (ICCARP) [2] have banded together to write this review, aiming to categorize the age-related diseases of the heart.
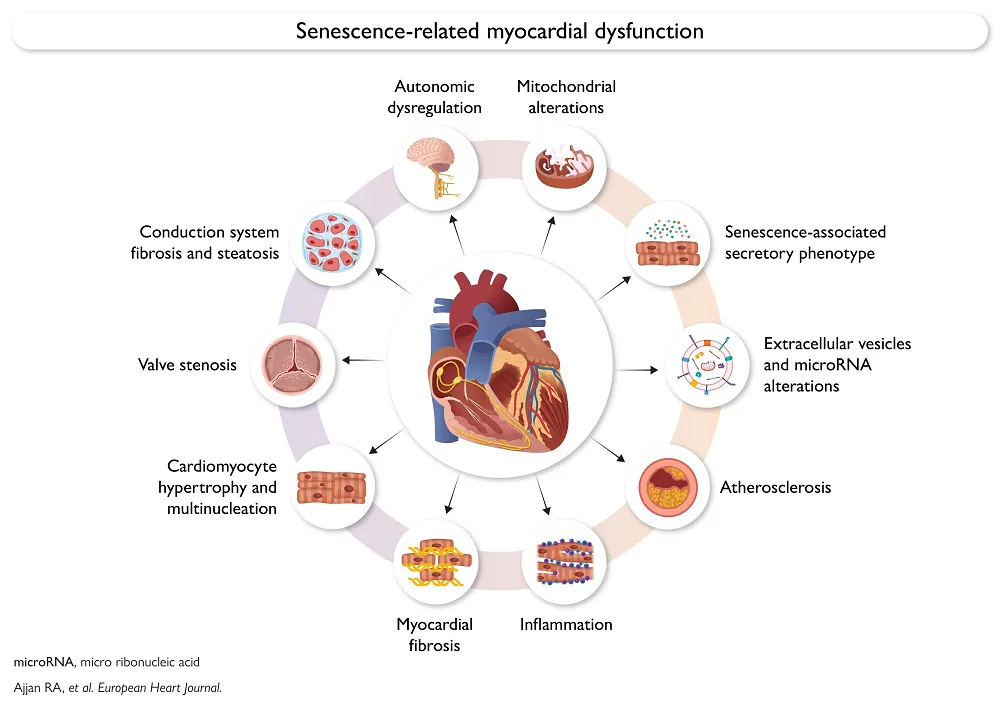
Cellular changes
Functional heart muscle cells (cardiomyocytes), which comprise 30% to 50% of the heart’s cells but most of its size [3], normally do not decrease in number with age outside of cardiovascular disease [4], despite the heart losing mass with age [5]. While the mechanism of new cardiomyocyte formation has not been directly observed in adult humans, they are most often directly generated from existing cardiomyocyte division, rather than from stem cells, in mice [6].
However, these cells are known to become senescent, which impairs their ability to function. This puts more workload on the healthy cardiomyocytes, leading to hypertrophy [7], and these cells secrete the well-known inflammatory factors common across senescent cells, causing further problems [8].
Senolytics, treatments that kill senescent cells, have been found to have beneficial effects against cardiac senescence in mice [9]. However, one of the most powerful, navitoclax, may affect how stem cells transition and may not be beneficial for overall mortality in this case [10]. Senomorphics, which modify how senescent cells function, have been also found to have some benefits, such as reducing the production of reactive oxygen species (ROS) along with fibrosis and cellular death by apoptosis [11].
Oxidative stress and other mitochondrial issues
Mitochondria naturally generate ROS, and the resulting oxidative stress is a key aspect of aging in the heart. It causes mitochondrial DNA (mtDNA) to oxidize [12] and mitochondria to swell [13], leading to functional impairment. It may be possible to deal with this mitochondrial swelling by administering sirtuins, and this approach has been found to alleviate hypertrophy in mice [14].
Mitochondrial dysfunction in the heart also manifests in other forms, with reductions in crucial forms of cellular respiration. While the enzymatic activities of the four main mitochondrial supercomplexes were found to be unaffected with age in the human heart [15], work in rats found that older heart tissue simply produces less of them [16]. Unsurprisingly, this coincides with a reduction in the energy currency ATP [17].
There are also alterations to how cardiomyocytes process food energy. Fatty acid oxidation decreases, resulting in an increase in glucose metabolism instead [18]. This leads to lipid accumulation inside cells, which is associated with diabetes [19] and a decrease in function [20].
Fibrosis, blood flow, and systemic problems
The sinoatrial node, which governs the pacemaking ability of the heart, is altered substantially with age. After fats and elastic tissue have infiltrated the area, this natural pacemaker of the average 75-year-old is completely different from that of the average 30-year-old [21]. Unsurprisingly, these changes, along with an increase in overall fibrosis that affects the heart and many other organs with age, lead to arrythmias and explain why artificial pacemakers are commonly used [22]. This increase in fibrosis is linked to an overproduction of matrix metalloproteinases (MMPs) and collagens, while their related inhibitors are reduced [23].
The heart needs its own blood supply to function, and losing this supply is the definition of a heart attack. Angiogenesis, the process by which the body produces more blood vessels, is reduced with age, contributing to the problem [24]. Blood vessel blockages known as ischemias, which cause heart attacks and strokes, are well-known in the cardiac context but, as they occur throughout the circulatory system, are not exclusive to any one organ.
Apart from direct aging of the heart’s basic systems, the researchers mention other systemic issues that cause heart aging. Immune cell dysfunction can lead to senescence being exacerbated, and inflammaging, the age-related increase in overall inflammation in the absence of pathogens, is also a key factor in the heart [25]. Similarly, circulating extracellular vesicles can be beneficial or harmful.
The reviewers also note that not all age-related changes to the heart are harmful. Hypertrophy itself, for example, is a reaction to damage; trying to directly address hypertrophy without addressing the underlying causes of senescence would not be beneficial. The reviewers also suggest that the lipids and fatty tissues that accumulate in the natural pacemaker serve a similar purpose.
Overall, there are several heart-specific and systemic issues that must be addressed if this crucial organ is to completely treated for age-related diseases. While its function of pumping blood may be relatively simple, its internal workings distinctly are not, and each of them will need to be dealt with if medicine is to substantially reduce heart failure in older people.
Literature
[1] van Riet, E. E., Hoes, A. W., Wagenaar, K. P., Limburg, A., Landman, M. A., & Rutten, F. H. (2016). Epidemiology of heart failure: the prevalence of heart failure and ventricular dysfunction in older adults over time. A systematic review. European journal of heart failure, 18(3), 242-252.
[2] Short, E., Calimport, S., & Bentley, B. (2025). Defining an ageing-related pathology, disease or syndrome: International Consensus Statement. GeroScience, 47(2), 1713-1720.
[3] Litviňuková, M., Talavera-López, C., Maatz, H., Reichart, D., Worth, C. L., Lindberg, E. L., … & Teichmann, S. A. (2020). Cells of the adult human heart. Nature, 588(7838), 466-472.
[4] Bergmann, O., Zdunek, S., Felker, A., Salehpour, M., Alkass, K., Bernard, S., … & Frisén, J. (2015). Dynamics of cell generation and turnover in the human heart. Cell, 161(7), 1566-1575.
[5] Olivetti, G., Melissari, M., Capasso, J. M., & Anversa, P. (1991). Cardiomyopathy of the aging human heart. Myocyte loss and reactive cellular hypertrophy. Circulation research, 68(6), 1560-1568.
[6] Senyo, S. E., Steinhauser, M. L., Pizzimenti, C. L., Yang, V. K., Cai, L., Wang, M., … & Lee, R. T. (2013). Mammalian heart renewal by pre-existing cardiomyocytes. Nature, 493(7432), 433-436.
[7] Suda, M., Paul, K. H., Minamino, T., Miller, J. D., Lerman, A., Ellison-Hughes, G. M., … & Kirkland, J. L. (2023). Senescent cells: a therapeutic target in cardiovascular diseases. Cells, 12(9), 1296.
[8] Evangelou, K., Vasileiou, P. V., Papaspyropoulos, A., Hazapis, O., Petty, R., Demaria, M., & Gorgoulis, V. G. (2023). Cellular senescence and cardiovascular diseases: moving to the “heart” of the problem. Physiological reviews, 103(1), 609-647.
[9] Walaszczyk, A., Dookun, E., Redgrave, R., Tual‐Chalot, S., Victorelli, S., Spyridopoulos, I., … & Richardson, G. D. (2019). Pharmacological clearance of senescent cells improves survival and recovery in aged mice following acute myocardial infarction. Aging cell, 18(3), e12945.
[10] Sutanto, H., Fetarayani, D., Narendra, M. R., & Nasution, S. A. (2025). The role of the senescence-associated secretory phenotype in cardiovascular disease among the elderly. European Journal of Internal Medicine, 106488.
[11] Zheng, H., Li, T., Hu, Z., Zheng, Q., & Wang, J. (2024). The potential of flavonoids to mitigate cellular senescence in cardiovascular disease. Biogerontology, 25(6), 985-1010.
[12] Hayakawa, M., Hattori, K., Sugiyama, S., & Ozawa, T. (1992). Age-associated oxygen damage and mutations in mitochondrial DNA in human hearts. Biochemical and biophysical research communications, 189(2), 979-985.
[13] Lesnefsky, E. J., Chen, Q., & Hoppel, C. L. (2016). Mitochondrial metabolism in aging heart. Circulation research, 118(10), 1593-1611.
[14] Hafner, A. V., Dai, J., Gomes, A. P., Xiao, C. Y., Palmeira, C. M., Rosenzweig, A., & Sinclair, D. A. (2010). Regulation of the mPTP by SIRT3-mediated deacetylation of CypD at lysine 166 suppresses age-related cardiac hypertrophy. Aging (albany NY), 2(12), 914.
[15] Miró, Ò., Casademont, J., Casals, E., Perea, M., Urbano-Márquez, Á., Rustin, P., & Cardellach, F. (2000). Aging is associated with increased lipid peroxidation in human hearts, but not with mitochondrial respiratory chain enzyme defects. Cardiovascular research, 47(3), 624-631.
[16] Gómez, L. A., Monette, J. S., Chavez, J. D., Maier, C. S., & Hagen, T. M. (2009). Supercomplexes of the mitochondrial electron transport chain decline in the aging rat heart. Archives of biochemistry and biophysics, 490(1), 30-35.
[17] Okada, M., Mitsunami, K., Inubushi, T., & Kinoshita, M. (1998). Influence of aging or left ventricular hypertrophy on the human heart: contents of phosphorus metabolites measured by 31P MRS. Magnetic resonance in medicine, 39(5), 772-782.
[18] Kates, A. M., Herrero, P., Dence, C., Soto, P., Srinivasan, M., Delano, D. G., … & Gropler, R. J. (2003). Impact of aging on substrate metabolism by the human heart. Journal of the American College of Cardiology, 41(2), 293-299.
[19] Zhang, L., Ussher, J. R., Oka, T., Cadete, V. J., Wagg, C., & Lopaschuk, G. D. (2011). Cardiac diacylglycerol accumulation in high fat-fed mice is associated with impaired insulin-stimulated glucose oxidation. Cardiovascular research, 89(1), 148-156.
[20] Van Der Meer, R. W., Rijzewijk, L. J., Diamant, M., Hammer, S., Schär, M., Bax, J. J., … & Lamb, H. J. (2008). The ageing male heart: myocardial triglyceride content as independent predictor of diastolic function. European heart journal, 29(12), 1516-1522.
[21] Davies, M. J., & Pomerance, A. (1972). Quantitative study of ageing changes in the human sinoatrial node and internodal tracts. British heart journal, 34(2), 150.
[22] Feinberg, W. M., Blackshear, J. L., Laupacis, A., Kronmal, R., & Hart, R. G. (1995). Prevalence, age distribution, and gender of patients with atrial fibrillation: analysis and implications. Archives of internal medicine, 155(5), 469-473.
[23] Biernacka, A., & Frangogiannis, N. G. (2011). Aging and cardiac fibrosis. Aging and disease, 2(2), 158.
[24] Rivard, A., Fabre, J. E., Silver, M., Chen, D., Murohara, T., Kearney, M., … & Isner, J. M. (1999). Age-dependent impairment of angiogenesis. Circulation, 99(1), 111-120.
[25] Młynarska, E., Kowalik, A., Krajewska, A., Krupińska, N., Marcinkowska, W., Motor, J., … & Franczyk, B. (2025). Inflammaging and senescence-driven extracellular matrix remodeling in age-associated cardiovascular disease. Biomolecules, 15(10), 1452.