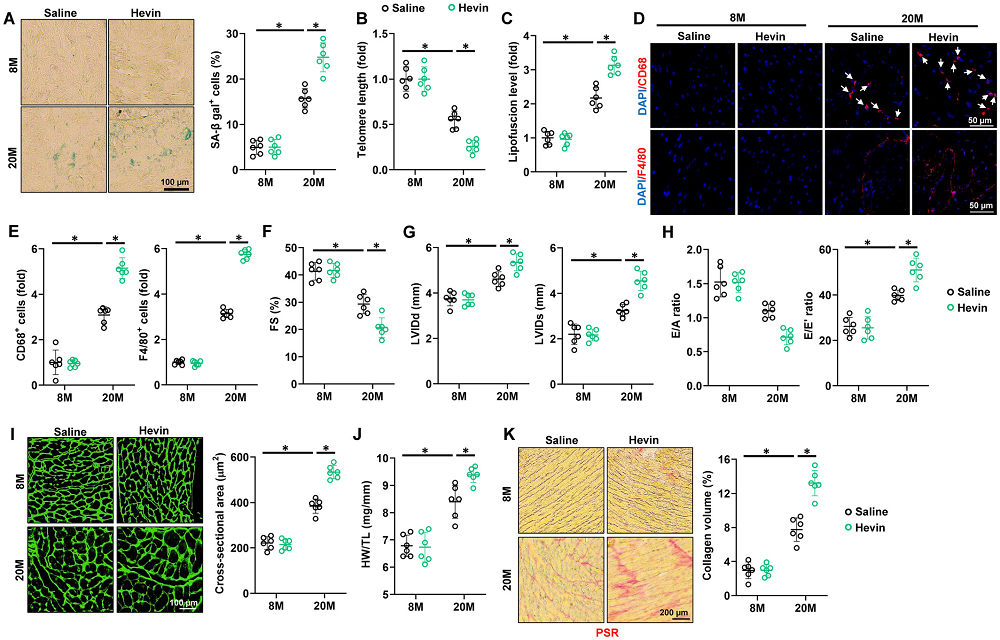
The last installment in our end-of-year series of expert roundups might be the least flashy, but it is arguably no less important than the previous ones dedicated to longevity biotech and geroscience.
Public advocacy is a powerful lever. To quote Abraham Lincoln: “Public sentiment is everything. With it, nothing can fail; against it, nothing can succeed.”
Wide public support could turn longevity into the next global moonshot project, with governments dedicating immense resources and creating a favorable regulatory climate, while the public pushes for faster change. Currently, we are far from this point, but the tide might be turning.
We asked six prominent figures in longevity advocacy to share their thoughts on the movement’s ups and downs in 2025 and its prospects going into 2026.
What successes and failures did longevity advocacy have in 2025, and how did they change your long-term outlook on its prospects?
Andrew Steele, author of Ageless, longevity scientist and advocate
I think 2025 continues a trend we’ve seen over the last few years which is both good and bad: the word ‘longevity’ has become increasingly visible, and concepts like biological age are being discussed more both on social media and in traditional newspapers, magazines, and so on. The good news is that the idea of measuring and even slowing biological aging is catching on. The bad news for longevity science is that it’s mostly a repackaging of well-worn lifestyle advice – sleep well, get some exercise, eat whatever diet the newly pivoted ‘longevity’ influencer was promoting before, and so on – and doesn’t touch on the idea of medical interventions to slow aging, which is where the real prize is.
Melissa King and Bernard Siegel, co-founders of the Healthspan Action Coalition (HSAC)
2025 marked longevity’s shift from marginal science to mainstream policy concern. States tested the boundaries of access and regulation. Montana expanded “Right to Try” beyond terminal illness, and Florida legislation increased access to unapproved stem cell therapies, but both demand scrutiny for patient safety and efficacy.
The THRIVE Act (Therapeutic Healthspan Research, Innovation, and Validation Enhancement Act), proposed U.S. legislation, was spearheaded by the Kitalys Institute and HSAC to create a new FDA regulatory pathway for healthspan-extending products, incentivizing treatments that improve the years lived in good health rather than just extending lifespan. This act aims to overcome current barriers by offering incentives like market exclusivity for therapies targeting aging-related diseases, making healthy longevity a national health priority.
Washington has, in Lifespan.io’s words, “arguably the most pro-longevity administration in history,” through key appointments, while pharma committed billions to longevity programs. However, proposed budget cuts for NIH and NSF budget cuts threaten critical basic research. Broad institutional and patient advocacy coalitions likely will prevail keeping the budgets close to being fully funded. More problematic is a loss of expertise and experience in Institute leadership.
Commercialization setbacks, including Unity Biotechnology’s shutdown and Calico partnership dissolution, exposed the field’s uncertainty. Meanwhile, premature clinic proliferation, inconsistent biomarker validation, and high-cost interventions risked eroding public trust and deepening equity gaps.
Our outlook is optimistic. Political recognition creates unprecedented regulatory opportunities, but funding crises and credibility risks could close this window quickly. The field must prioritize rigorous evidence, standardized protocols, and equitable access, or risk squandering a historic moment. The infrastructure we build now determines whether longevity becomes a transformative public health policy or boutique medicine for elites.
Dylan Livingston, founder and CEO of the Alliance for Longevity Initiatives (A4LI)
2025 felt like a turning point. The biggest signal to me was getting someone like Jim O’Neill [O’Neill is a former CEO of SENS Research Foundation] into the Deputy Secretary of HHS role; that’s a level of institutional leverage the field hasn’t had before.
Some people expected overnight change, and that was the main “failure” of our industry in 2025: unrealistic timelines and impatience. Biotech and policy compound slowly, then quickly. My long-term outlook is more bullish, not because everything changed instantly, but because the right levers are now closer to being pulled – and once one major barrier moves, a lot of other things can move behind it. I also think Jim’s appointment set a precedent by making it easier for future administrations to bring longevity-literate leaders into senior roles and treat this as a serious policy priority.
Adam Gries, co-founder of the Vitalism Society
2025 marked a shift from aspiration to law. States led the charge: New Hampshire’s HB 701 substantially expands right-to-try for terminal patients; Montana’s SB 535 created a licensed framework for experimental treatment centers using post-Phase I therapies, positioning the state as a potential hub for innovation. Florida’s stem cell carve-out (effective July 1) allowed physicians to administer certain non-FDA-approved therapies under rigorous manufacturing and consent rules – controversial, but a clear signal of demand for faster translation.
Federally, Dylan Livingston and A4LI grew the bipartisan Longevity Science Caucus significantly, with Dr. Mehmet Oz headlining their fly-in and lending real political momentum.
Some gaps remain. We still lack a pathway for prevention-focused biologics, and the community hasn’t encouraged enough great scientists and innovators into government roles.
My outlook is unchanged and bullish. Advocacy is still the cheapest, highest-ROI lever for change, especially with overall longevity-positive tailwinds.
Anastasia Egorova, CEO of Open Longevity
If we measure success by public recognition, then yet again, no one outperformed Bryan Johnson. However, that is a very niche type of fame, and the jury is still out on whether it actually benefits longevity R&D – you know, the science that will actually extend our lifespans.
I know of several projects initiated in 2025 that went barely noticed, even by the longevity community itself (a couple of new books, a new documentary in the making, and so on). Let’s count them as seeds planted for the future; they have potential, even if they’re quiet.
As for advocacy’s influence on policy and funding? It’s too early to call. The year felt like one long transition period; this world moves slowly.
In summary: there were no obvious wins for advocacy in 2025, but R&D itself isn’t looking too bad. I’d say the probability of success within our lifetimes (specifically for those currently 35-55) shifted from 0.1% to… let’s say, 0.1-something-percent. It’s movement.
Which event from 2025 – in politics, policy, public opinion, media, or popular culture – do you consider the most influential for longevity advocacy, and why? You’re welcome to name a runner-up.
Andrew Steele
I think, frustratingly, that the biggest longevity story of 2025 was the exchange between Chinese and Russian presidents Xi Jinping and Vladimir Putin in September, where they were caught on mic discussing living to 150 through repeat organ transplantation. This was heavily covered by global media and played into multiple negative stereotypes about longevity science: that it will mean dictators can hold onto power for longer, that the medicines will only be available to the rich, and so on. This is another reason longevity science needs a stronger voice in popular culture; none of the coverage I saw pushed back on these stereotypes, and it would be great if the positive sides of longevity got an airing too after an event like this.
Melissa King and Bernard Siegel
For HSAC, the core takeaway is not technological acceleration but governance. 2025 is the year that longevity entered the public policy arena but before it earned durable public legitimacy. Longevity is no longer just a scientific or commercial conversation. It is now a question of public responsibility. Who gets access, under what standards of evidence, and with what protections against harm and inequity are all questions that must be answered.
Our role is to help ensure that this transition strengthens public trust and does not undermine or seek to abuse it. The field advanced, and continues to advance, faster than its regulatory, ethical, and equity frameworks. Without coordinated action, early policy wins risk being interpreted, and even implemented, as deregulation rather than public health progress.
Dylan Livingston
Since I already mentioned Jim, I’ll be a shameless self-promoter and say A4LI’s Montana Right-to-Try bill. It put longevity on the map in a really meaningful way. We’ve never had longevity legislation written about in the Wall Street Journal and other major outlets, and that matters because it shows two things at once: first, the industry is making real progress, and second, the mainstream is starting to take longevity seriously.
Runner-up: the continued acceleration of the science and the companies behind it – more trials moving forward, more funding, and more real momentum.
Adam Gries
Hands down, the most influential was the appointment of key longevity-friendly leaders: Jim O’Neill as Deputy HHS (a longtime advocate who ran SENS Research Foundation), Dr. Oz at CMS, Marty Makary at FDA, and Alicia Jackson at ARPA-H.
All have publicly framed aging as malleable and are positive towards longevity science. This makes the current administration the most pro-longevity in U.S. history. The respective roles oversee NIH funding, FDA approvals, Medicare/Medicaid policy, and high-risk research, directly impacting translation of aging biology into therapies.
Collectively, these shifts move longevity to the mainstream of public priority.
Anastasia Egorova
Like most other causes, longevity advocacy is at the mercy of macro-politics. We have a new administration in the country responsible for the bulk of longevity R&D, and we have wars burning through resources and distracting the public. Worse, these conflicts harden the collective psyche, making people even more prone to justifying death. The value of human life, economic stability, global optimism – all of these affect our field. The problem is we still don’t have good metrics for this influence and public attitudes towards longevity in general.
On the media front, MIT Technology Review ran two bold ethics-related articles on a topic that was tabooed until recently: “Spare” living human bodies might provide us with organs for transplantation” and “Ethically sourced “spare” human bodies could revolutionize medicine.” Then we had that viral clip of Putin and Xi discussing immortality and organ transplantation. That is certainly publicity, but did it actually help the science of organ replacement? Again, we don’t have a way to measure it.
AI might help with both the activism and measuring its effects. We just need more actual human talent in longevity advocacy.
Thinking back to what you believed about the state of longevity advocacy on January 1, 2025: did anything in 2025 genuinely surprise you or make you update your priors?
Andrew Steele
I think the most surprising thing was when my dentist asked me about my appearance in Don’t Die, the Bryan Johnson documentary that came out on January 1, 2025! I live in Berlin, so that means I’m a step further removed from English-language longevity content, but apparently the message is getting through. To me, it was another sign of how fast the concept of longevity has moved in the last year and another example of the public receiving the message in a form that doesn’t always do longevity science favors.
Melissa King and Bernard Siegel
HSAC anticipated that longevity advocacy would advance through scientific validation first, then cultural acceptance, followed by policy change. We expected public institutions to wait for consensus on biomarkers, endpoints, and interventions before committing resources or political capital.
It surprised us how quickly policy moved, often ahead of scientific consensus. State-level Right-to-Try expansions and early federal proposals revealed political appetite for action, but not always for the guardrails needed to ensure safety, efficacy, and equity.
This updated our prior belief that policy would be guided by science. Instead, we saw policy move first, shaping the field for better or worse. Advocacy therefore cannot wait for scientific closure. Without early engagement, longevity risks being defined through deregulation rather than responsible governance.
We were struck by the fragility of public support for research. Proposed cuts to NIH, including the NIA, exposed a disconnect between growing interest in longevity and sustained investment in the science required to legitimize it, even as ARPA-H directed a significant share of its smaller budget toward longevity.
Most unexpectedly, credibility emerged as the field’s most immediate risk. Premature commercialization, inconsistent biomarker claims, and rapid clinic proliferation threatened public trust, while high-cost interventions increased, rather than decreasing, equity concerns.
Dylan Livingston
Not really. If you’ve tracked the field for years, you know the trajectory has been building. Biotech takes time, and a lot of the organizations and companies launched in the late 2010s and early 2020s are now reaching stages where tangible things start happening.
To a non-insider, the pace of clinical updates, funding, and legislation can look surprising, but I expected the tempo to increase around now. If you zoom out (Kurzweil-style timelines and all), this is roughly the window that many people have pointed to as the start of more visible acceleration toward longevity.
Adam Gries
The diversity and speed of state-level regulatory strategies. Bills like Montana’s SB 535, New Hampshire’s HB 701, and Florida’s stem cell expansions showed states willing to innovate, creating platforms for accelerated innovation and delivery of potential longevity therapies.
The key goal, which affects longevity therapies alongside all therapies is: how can we get therapies to humans faster and more cheaply without sacrificing safety.
While longevity advocacy remains highly underfunded, these outcomes prove targeted effort can yield outsized results. It updated my priors upward on how quickly regulatory momentum can build bottom-up when the science is compelling, and public interest in healthier aging grows.
Anastasia Egorova
[After some deliberation, Anastasia decided not to respond to this question.]
What are your expectations for 2026 in terms of opportunities and risks for longevity advocacy?
Andrew Steele
As a field, we need to work harder to own the narrative and reclaim the word ‘longevity’ for real longevity science. That’s why in 2025 I co-founded The Longevity Initiative with lawyer and longevity advocate Kamen Shoylev. We want to bring real longevity research to policymakers, scientists, doctors and the public, and be a part of recapturing ‘longevity’ for real lifespan- and healthspan-extending research. We’re starting out in the UK and Europe, not least because there are already excellent organizations like the Lifespan Research Institute with more of a US focus, and we look forward to working together to spread this vitally important message in 2026.
Melissa King and Bernard Siegel
We believe that healthspan should be declared a fundamental human right. In 2026, this global advocacy campaign will begin in earnest. Promotion of this lofty goal will unify stakeholders, galvanize public discourse, and bring healthy longevity, and the benefits of converging technologies, to all populations.
2026 will be a year of institutionalization. Longevity advocacy has an opening to move from pilot policies and exploratory legislation into more durable structures, including regulatory pathways, public-sector research commitments, and early reimbursement conversations.
The federal interest in healthspan, combined with continued pharmaceutical and biotech investment, creates a rare chance to align innovation with public health goals. For advocates, this is the moment to shape standards for evidentiary thresholds, biomarker validation, post-market surveillance, and equity safeguards. 2026 can be the year we start anchoring longevity within mainstream health policy rather than exceptional access frameworks.
The primary risk is overreach. If access expands faster than evidence, or if credibility erodes through premature commercialization and uneven clinical practices, public trust could collapse. Funding instability, especially at the US federal level, remains a structural threat. Equity risks will also intensify as high-cost interventions reach the market without clear pathways for broad access.
2026 will reward disciplined advocacy. Progress will depend less on breakthrough science than on whether the field proves it can govern itself responsibly in the public interest. If all goes well, the world will be changed for the better.
Dylan Livingston
More of the same, in the best way: more readouts, more new companies, more funding, and more foundational science translating into programs people can actually point to. The risk is PR and narrative warfare. As we get closer to real-world access and real-world policy, opposition gets louder and more creative. Our polling showed that when you explain longevity in plain language, people are broadly supportive. But the SB 535 testimony showed the other side of the coin: if you don’t control the framing, a loud minority can hijack the narrative, strip out the context, and replace it with misinformation.
In 2026, that means advocates need to fight on messaging as hard as we fight on policy – tight talking points, fast response, and repeatable, values-based framing that ties longevity to what people already want: more healthy years, more independence, less disease, and more dignity as we age.
Adam Gries
We need to encourage great talent to join the public sector, especially at NIH, FDA, CMS, and ARPA-H. States will keep innovating on regulation. We’ll see more proof-of-concept in accelerated translation, real-world data collection, and treatments. Also, growing the Longevity Caucus and securing more legislative support will build lasting momentum.
Risks include failing to attract top talent or pushing poorly scoped initiatives without considering the full set of stakeholder incentives. Progress could also be slowed if longevity gets politicized instead of staying bipartisan. With the right thoughtfulness and focus, we have a rare window to see rapid acceleration of meaningful aging science.
Anastasia Egorova
The biggest risk for longevity advocacy is irrelevance. In fact, this is, sadly, our starting point: we ARE currently mostly irrelevant. Compared to mainstream politics or entertainment, we barely register.
But therein lies some good news, too. If you have ideas and an appetite for longevity advocacy, the bar is low. Think about it: your biggest competition is a guy swapping plasma with his son. I have the utmost respect for Bryan and his team; it just shows that the field is wide open. So, be bold. Do whatever you want. Connect with others. Launch projects. Break boundaries.
We would like to ask you a small favor. We are a non-profit foundation, and unlike some other organizations, we have no shareholders and no products to sell you. All our news and educational content is free for everyone to read, but it does mean that we rely on the help of people like you. Every contribution, no matter if it’s big or small, supports independent journalism and sustains our future.