Dr. Maximina Yun, principal investigator at Chinese Institutes for Medical Research in Beijing (CIMR), studies some of the most amazing animals in the world: salamanders, a group of amphibians that includes newts and species such as the universally loved axolotl. On top of being cute, salamanders possess unparalleled regeneration abilities for vertebrates, being able to regrow organs and limbs.
Salamanders are also notoriously long-lived for their body sizes, with axolotls hitting around 20 years while demonstrating negligible senescence and cave olms sporting a mind-boggling maximal lifespan of over 100 years. Studying salamanders is not easy, but the potential rewards are enormous. We talked to Maximina about her interest in these animals and the ways that we can utilize their phenomenal adaptations for ourselves.
How did you become a geroscientist, and what does studying human aging mean to you personally?
I arrived in this area because of my interest in DNA repair. I did my PhD on mechanisms of genome stability and maintenance, and I had a strong interest in cellular plasticity. That is what brought me to do a postdoc in Jeremy Brockes’ lab and begin to understand the mechanisms of cellular plasticity commonly used during salamander regeneration.
Eventually, working with this model, I realized that there is a strong potential for using salamanders to understand the links between regeneration and aging. One of the reasons is that salamanders have long been considered organisms of negligible senescence. This is largely based on mortality studies indicating that the rate of death does not increase with age in salamanders, and actually this is seen in all species studied so far.
Moreover, if you go into the very early literature, you will find reports stating that we are unable to determine a salamander’s age. There is no such thing as differentiating a newt that is one year old or two years old versus one that is twenty years old. That presented a lot of potential, and I became interested from the biological side. Obviously, aging is a pressing challenge – probably the biggest challenge of the 21st century. But for me, as a biologist, what really pulled me into the field was the potential link with cellular plasticity and understanding how these two big processes, regeneration and aging, interplay.
I think salamanders are a phenomenal model. Tell me more about them – their regeneration abilities, the negligible senescence, and all the things they can do that we can’t.
Salamanders are a very special type of organism. They are amphibians, very close to Xenopus (frogs). But while Xenopus loses its regenerative ability through adulthood, salamanders keep it regardless of whether they go through metamorphosis or not. All salamander species reported so far are known to regenerate structures. It is thought that the ancestor of salamanders was able to regenerate. Actually, there is an area in Germany called Pfalz which has fossil records of ancient salamanders at different stages of regeneration. My postdoctoral mentor, Jeremy Brockes, used to have a couple of such fossils in his office and they never failed to leave me in awe.
This ability is remarkable among vertebrates. Particularly, they are the only tetrapods able to regenerate their nearly-full limbs as adults. They can regenerate parts of their brains – in experiments with newts, if you remove half the optic tectum, it will grow back. It takes a long time, over six months, but it will grow back.
They are also able to regenerate their ovaries, and work from my talented student Yuliia Haluza looking at thousands of matings over 15 years in the Dresden axolotl colony indicates that the axolotl retains fertility through lifespan, in keeping with their extreme aging resilience.

Axolotl at Vancouver Aquarium. Photo: Arkadi Mazin
They can also regenerate up to a third of their heart, their tail including the spinal cord, maxillary bones, gills. This is really remarkable. They even regrow structures they don’t necessarily need. For example, the axolotl never undergoes metamorphosis, but it can regrow its lung even though it’s never going to use the lung for breathing on land. It’s not selected for utility, which is interesting. It’s an example of residual regeneration.
They are powerful at regenerating, but there is a catch: in all known cases of regeneration – except for the newt lens, which is a particular example – you always needed the remnant of the structure in order to regrow it. However, our lab has just discovered that the limits of regeneration can be extended further: axolotls are able to regrow their thymus de novo. This means they can regrow their thymus completely from scratch, and it’s the one example of a complex organ that can be fully regrown among vertebrates! This is truly exciting, as it present a completely different regeneration paradigm and further highlights the power of this model for regeneration research.
With regards to ageing, this raises key questions too: does the thymus involute with time in axolotls, as it does in mammals? Is its age-related homeostasis enhanced due to the regenerative abilities? Largely, how does a super regenerator age? Many exciting avenues ahead of us.
When they regenerate their brains, are memory and learning preserved?
We don’t know yet. Until recently, salamanders were not fully experimentally tractable. For example, the axolotl genome only came out in 2018, and the first chromosome assembly for the Spanish ribbed newt – the most tractable newt model – came out just in 2025, an effort led by the labs of Nick Leigh, Andras Simon and mine. Compared with other model organisms, this is all too recent. However, it means we can now truly exploit the wide range of tools existing for more traditional systems. There are memory paradigms currently being developed by a few research groups, both for axolotls and newts, and we are all looking forward to the results.
I think their genome is about ten times larger than the human genome, right?
It is ten times larger. There were a lot of complicatons because most salamanders exhibit genomic gigantism. Largely due to massive expansions of repetitive elements, not genome duplication (actually, the axolotl and the Iberian ribbed newt are both diploid). As they are extremely large and highly repetitive, standard sequencing approaches did not work well. Recent advances in genome sequencing tech, particularly PacBio long-read sequencing, made this possible.
So, for geroscience, it’s a fairly new model, and they also usually have long lifespans, correct?
That’s true. Although different salamanders have very different lifespans. For axolotls, the average is 10 to 13 years of age while their maximum lifespan is about 20 or 21. Newts are significantly longer lived, and then cave olms, such as Proteus anguinus, live well over 100 years! They are all lifespan outliers based on their body size.
But we don’t know for sure, say, from your colonies?
It is in fact from our colonies! We had a 21-year-old, one of the longest-lived axolotls. When the model is so long-lived, it’s difficult to do the studies. With newts for example, there have been a lot of capture-recapture studies in the wild. Critically, whenever a salamander species has been studied in terms of lifespan, one thing is obvious: there is no increase in mortality rate with age.
So, that’s negligible senescence.
Exactly. We classify them as having negligible senescence because this has been tested in several species and none of them exhibit mortality increase with age. But nothing is known at the molecular level, and this is where our lab jumps in: in one huge project, my lab is characterizing how these animals age, and how this is different from senescent species such as us.
I’m wondering if there is a relationship between regeneration and negligible senescence in these species.
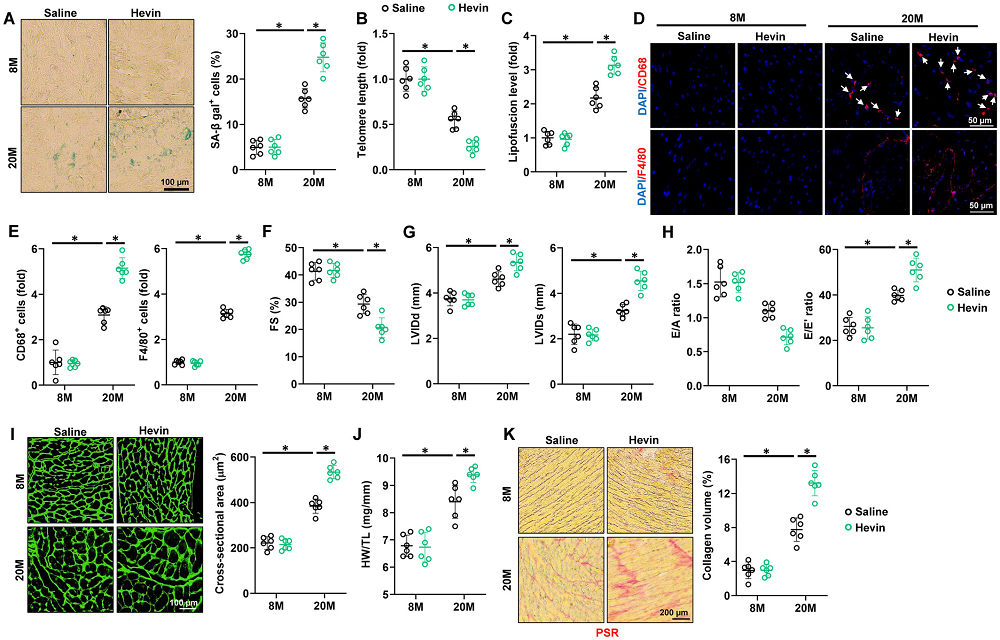
That’s exactly what we are working on right now. The first attempt to address this was through building epigenetic clocks, together with Steve Horvath’s team. We found that we can build an epigenetic clock for up to four years of aging in the axolotl, which is the early life. Past that point, it seems to stabilize epigenetically, a phenomenon we have never seen before and could relate to negligible senescence traits.
With the part of the clock that works after four years of age, we used this to ask what happens to limbs or tails which have undergone several regeneration cycles. The answer is that if you compare a forelimb that has regenerated three times versus the contralateral limb that has never regenerated, the one that has regenerated three times is epigenetically younger by DNA methylation age than the contralateral limb.
This suggests there might be some rejuvenation events associated with regeneration, which is exciting because it’s in a natural setting. This organism does it naturally; it’s not reprogramming with Yamanaka factors, and it raises multiple questions which we’re trying to address. The epigenetic and other tools we are currently developing will help providing important answers with regards to the interplay between regeneration and aging.
If I remember correctly, regeneration in forlimbs is linked to cellular senescence.
Cell senescence is elicited every time you regenerate a limb. It appears at particular stages during regeneration, and it contributes to fueling the process, particularly by secreting factors – for example, Wnt signals – that will promote plasticity (in newts) or proliferation of the neighboring cells (in axolotls).
As regeneration progresses, you cannot have these processes turned on forever. We have seen that the number of senescent cells starts to decline as the limb progresses through regeneration. This depends on clearance by the immune system, in particular the macrophages.
We know that senescent cells play a role in wound healing in mammals. Is this some sort of a remnant of this mechanism?
It’s not clear. If you read Marco Demaria’s work – he and the late Judith Campisi uncovered the roles of senescence in wound healing – they are acting by secreting PDGF factors that promote the more rapid migration of the cells to close the wound. This is a different mechanism from what we see in salamanders, which fuels formation and proliferation of the blastema progenitors.
We don’t know exactly if they are related. Whether there are commonalities between this context of “beneficial” senescence that are different from the “negative,” pro-inflammatory senescence you see in aging is a very interesting question. We would love to explore this more with others in the field.
Salamanders also appear to have a superior clearance mechanism for senescent cells, right?
The clearance we see is very effective. Not only do the dynamics of regeneration lead to regenerative limbs that have no senescent cells – you can try to find them, but you won’t – but they never linger. Not even a couple, which is remarkable. We know that macrophages are required for their clearance, but little beyond this. There is a great opportunity here to probe the basis of efficient clearance mechanism, which I am convinced could inform strategies to improve clearance in mammalian systems.
Senescent cells are proposed to play roles in limb development, is it also the case in salamanders?
This is also different from development. If you see a developing limb bud, you won’t see senescent cells, but if you induce regeneration in that developing limb bud, then you will see senescent cells. I sometimes get asked if regeneration is similar to development. I would say it is not a full recapitulation. There are many differences. Senescent cell intervention and usage is different, and there are many other aspects. Of course, some of the patterning programs used to regrow the structure are conserved between development and regeneration, but there are many differences, especially at the beginning of the process.
There’s also dedifferentiation, maybe transdifferentiation of cells during regeneration. It’s a really interesting phenomenon, something like cell reprogramming in vivo.
What we know is that salamanders are masters at regulating cellular identity and plasticity. The cells know very well what they are, and when they go back, they never forget what they are. When a limb regenerates, connective tissue only gives you connective tissue; muscle only gives you muscle.
For example, in the newt, the muscle is generated by dedifferentiation. Even though the muscle comes from dedifferentiation, the resulting regenerative progenitors never fully lose their muscle marks and they only give you muscle.
Experiments with genetic tracing, single-cell analysis, and transplantations tell you the specific tissues overall maintain their identity. Dedifferentiation is also critical for axolotl limb regrow, whereby the connective tissue cells dedifferentiate to give you all CT-derived tissues. This is one of the most important principles when these animals regenerate.
Transdifferentiation is another mechanism these animals use. For example, in the newt lens: you can remove the lens of the eye completely, and the entire lens will come back by a transdifferentiation of the dorsal pigmented epithelial cells from the iris. When you remove the lens, these pigmented epithelial cells undergo transdifferentiation to form crystalline cells that form the lens. This is one example where pure transdifferentiation is used.
But in the context of many other complex structures, we haven’t seen much of this transdifferentiation. There are recent reports, such as an exciting preprint from Wouter Masselink, Elly Tanaka and Prayag Murawala, which indicates that the tail mostly regenerates via specific stem-like progenitors. These stem cell-like cells can give you different tissues in the tail, but it’s a bit different; it’s like playing with the potency of a stem cell, not necessarily direct transdifferentiation as in the lens case.
Your group is also working on thymus regeneration in salamanders. This is exciting since thymus involution clearly plays a role in human aging.
It is really amazing because, first, it’s something you can see with your own eyes. The thymus in the axolotl sits at the base of the gills, formed by three bilateral nodules. You can remove the entire nodules including the connective tissue surrounding them, and, in six to eight weeks, the entire organ will be back. It’s a complex organ because it’s formed by thymic epithelial cells, thymic progenitors, hematopoietic progenitors, dendritic cells, endothelium, macrophages, and obviously the different lymphocytes, and everything just reappears.
We found that when these animals regenerate the thymus, they recapitulate the morphology, cell populations, and function. One can do these very cool experiments where you transplant a regenerated structure from one salamander – a thymus nodule from a salamander that is fluorescently labeled – into a [transparently] white host. Then you can see if this regenerated thymus can support the hematopoiesis and lymphopoiesis of the host.
You can see that it does, because after a year or two, this thymus nodule will still have its original thymic epithelial cells fluorescently labeled, but it will be supporting the entire thymopoiesis of the host. It’s wonderful.
How can we translate insights from these species into humans? What are the main hurdles? I must confess that every time I speak with someone about long-lived or regenerating species, I get envious. I immediately want to have those abilities.
The first thing is to do solid basic science that gets deep into cellular and molecular mechanisms. Understand what really underlies these remarkable traits, and only then you can generate a blueprint for guiding interventions.
Regarding hurdles and timescales, these are intertwined. Primarily, you need to consider the physiology and biological differences between species. Sometimes you may have a target that seems sensible, but it’s actually tied to the particular species’ physiology.
In the case of salamanders, they are ectotherms. They live at different temperatures and control their energy differently. If they’re adapted to live at 20 degrees, can we adapt their mechanisms for an organism that lives at 37 degrees? We have to think about all this. Sometimes you can try to replicate these interventions by playing with the organism’s own proteome. That’s great because you are not subject to these differences.
The biggest hurdle is: do we need one hit or ten hits to reproduce this? But that is just trial and error, leveraging datasets and doing preclinical studies. We have started to take some of our thymus findings into other models to see if we can promote thymus rejuvenation or delay involution by giving them these molecular features that the axolotl has. We’re already doing it, and it’s very exciting.
It’s great to know that you are already looking at the translational angle.
Very much so. This goes hand-in-hand with generating a solid fundamental basis.
What about the evolutionary aspect? Do we know why salamanders have preserved regenerative abilities and mammals lost them almost completely?
It is not necessarily clear in the field of regeneration whether regeneration is a conserved trait of all animals or an acquired trait and an example of convergent evolution. A number of scientists, including my postdoctoral mentor Jeremy Brockes, sustained the latter, supported by the fact that there are salamander-specific proteins involved in regeneration. It is quite possible that regeneration has evolved in salamanders in a different way than in other organisms. For example, if you look at Hydra or planarians, they regenerate in very different ways. It’s not that all organisms that regenerate use the same mechanisms. Different organisms have different solutions.
So, it’s not necessarily settled that we lost the ability to regenerate. There are still many views regarding the evolution of regeneration. Among them is the cancer connection – that organisms that regenerate very well would be more prone to cancer, but that’s not the case in salamanders. They’re actually very resistant to cancer.
They seem to have all the bases covered somehow.
Exactly. And if you think of salamanders as an organism, they occupy almost every niche. They are really evolutionarily successful. For example, you can find salamanders in Siberia. You have newts in the tundra that get frozen and stay dormant in a block of ice for long periods, but when the ice melts they just walk away. There’s a lot of adaptability there.
Obviously, humans are a threat to salamanders worldwide. The axolotl is a good example; in Lake Xochimilco, human activity resulted in water contamination from rapid urbanization and the introduction of carps into the system. They started to wipe out axolotl populations in the natural environment to the point of near extinction in their natural environment (though they exist in high numbers in pet shops and colonies!). Right now, there are efforts towards bringing them back, and we are all rooting for this.
Do you have an opinion on Michael Levin’s bioelectricity-focused research into regeneration?
Bioelectricity is very important. There were early studies in the sixties and seventies showing that reverting currents in the water prevents for example, newt limb regeneration. So, we know that bioelectricity plays an important role. It’s a way in which cells communicate. I think Mike’s work is very interesting. For the field, we want to see this expanded into how the different molecular networks impact bioelectricity and how this is coordinated during regenerative processes. It’s a very interesting angle.
You moved to China recently. How do you see China’s place in the modern geroscience landscape, and how is the research and biotech climate different from Europe?
As in many other sectors, China’s influence is growing at speed. The trend is for science to be heavily influenced by China soon. I think the government has made very right steps in investing in science and deciding China should become the next scientific powerhouse. The environment here is certainly very welcoming, increasingly so towards internationals.
The current international context has led to a significant import of foreign scientists, which are in turn strengthening China’s position worldwide. There are significant funding opportunities here, for both basic research and the biotech sphere, combined with the chance to tap into a pool of talented and super hardworking students and staff.
Things happen at a very fast pace, and the regulatory frameworks for experimentation are much more science-friendly than in Europe. There is still perhaps too much focus placed on high-impact publications, but there is a clear effort towards promoting rigorous science. I’ve been here for four months, so we’ll see how things develop, but what I’m experiencing so far is very exciting.
Do you see the agility that is often invoked with regard to China – things being done faster, more efficiently?
Things are being done much faster than anything I have ever experienced. Sometimes if you have too much speed, that can be counterproductive. But institutes like the one I am in, Chinese Institutes for Medical Research (CIMR), are set up with more than 90% of the faculty coming from Europe and the US, so it’s a bit different.
Personally, I was attracted by the idea conceived by Yi Rao and others to create an HHMI-style institute in Beijing. Here, the support system and evaluation processes are based on the success stories that happen in the West, modeling institutes like the Laboratory of Molecular Biology in Cambridge, where I did my PhD, and Janelia Farm.
One of the features of such places is also the possibility to have resources and time to explore great, bold ideas, and I feel this component is important for doing significant science. Overall, there is a view that this will lead to success here.
We would like to ask you a small favor. We are a non-profit foundation, and unlike some other organizations, we have no shareholders and no products to sell you. All our news and educational content is free for everyone to read, but it does mean that we rely on the help of people like you. Every contribution, no matter if it’s big or small, supports independent journalism and sustains our future.