Elevant launched its first product, Prime, in January 2021. Prime is a daily NMN supplement featuring a unique form of cGMP-manufactured NMN called NMN-C.
Guillaume Bermond is a French entrepreneur and founder of Elevant, an American health and wellness brand that develops cellular health solutions with leading scientists. Dr. Alessia Grozio is Elevant’s Chief Scientific Officer and a recognized leader in the field of NAD+ metabolism and aging.
First, congratulations on the recent launch of your product, Elevant Prime. How has the launch gone so far? How has its reception been among consumers and the scientific community?
GB: Thank you! Our first few months in-market with Elevant Prime have gone very well. We’ve been really pleased with the feedback we’ve received from customers. People are becoming more educated about NAD+ boosting, and NMN in particular, so they’ve responded very positively to the fact that we are making our own form of NMN (NMN-C) and producing Prime at cGMP facilities in Europe. Given the online nature of the cellular optimization industry, transparency and product accountability can be hard to find for consumers. The peace of mind we provide in those respects has struck a chord.
People have told us they are experiencing the benefits of supplementing with NMN in varying ways and degrees – which makes sense as, obviously, we’re all individuals and everyone’s biology is different. But we are receiving a lot of positive feedback about the effects people are feeling – elevated energy, mood stabilization, better sleep, and increased capacity for physical activity. We believe the product and timing is right for success as we are seeing people who usually take vitamin supplements telling us they are turning to Elevant, looking for something that works at a cellular level to support health and well-being.
AG: It’s been heartening to receive positive feedback (sometimes begrudgingly!) from industry players around our commitment to undertaking clinical research to further advance understanding and potential therapeutic uses of NMN. We are in a ‘take-no-prisoners’ industry, so to get that acknowledgement goes to show we are doing something right. Although recognition is nice, the motivator for us remains the benefit that more and more clinical evidence around NMN as a strategy to support healthspan can bring for people and communities.
Readers of lifespan.io are likely already familiar with NAD+ and the NAD+ precursor NMN, but for those who aren’t, can you give us an overview of the science behind this supplement? And what are the advantages of NMN as opposed to other NAD precursors?
GB: A key differentiator – and advantage – of NMN compared to other molecules marketed to consumers as NAD+ boosters is the biochemical processes involved. While other NAD+ precursors require conversion to NMN before finally converting to NAD+, NMN is the immediate precursor to NAD+ in what is called the ‘salvage pathway’. As the final step in the biochemical chain reaction that produces NAD+, NMN is a highly efficient route to generating NAD+ and, in turn, supporting vital molecular pathways that keep your body running well.
AG: Other NAD+ precursors have some characteristics that NMN does not (or if it does, it displays them to a much lesser extent).
For example, nicotinamide (NAM) does not efficiently increase NAD+ in cells because, to generate NMN, it uses nicotinamide phosphoribosyltransferase (NAMPT), which is a rate-limiting enzyme in the salvage pathway, meaning that NAMPT has a plateau of activity that can’t be overcome even if more NAM is provided.
Furthermore, NAM is also a byproduct of NAD+ catabolism. At millimolar (high) concentrations it has been shown to act as a feedback inhibitor for NAD+-dependent enzymes, such as PARPs and sirtuins (which are important for longevity) [1] [2], and to increase methylated NAM. High levels of methylated NAM have been associated with the pathogenesis of type 2 diabetes, Parkinson’s disease and cardiac diseases [3].
Nicotinamide Riboside (NR) is unstable in body fluids (blood and plasma) and it is degraded into NAM; therefore, all the caveats of NAM mentioned above can potentially be applied to NR [4] [5].
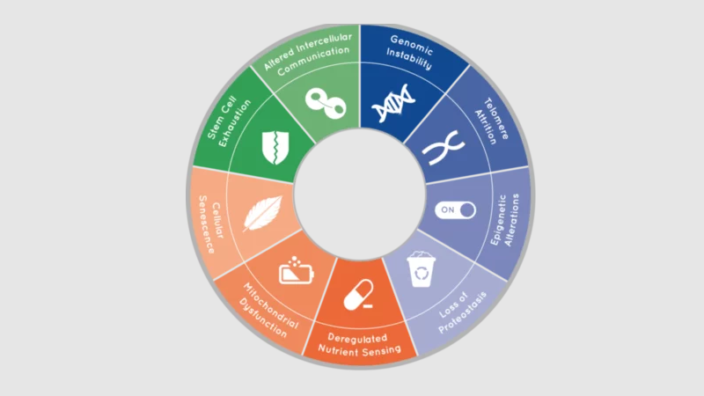
The Hallmarks of Aging is a common framework used to understand how interventions might modulate the aging processes. Which of these hallmarks has NMN been shown to impact and in what ways?
AG: NAD+ decline has been correlated with numerous hallmarks of aging, such as genomic instability, epigenetic alterations, telomere attrition, dysregulation of nutrient sensing, altered cellular communications, mitochondrial dysfunction, and compromised autophagy.
Boosting NAD+ levels via NMN administration has shown beneficial effects by promoting DNA repair (genomic instability), gene expression (epigenetic alterations), and oxidative phosphorylation and mitophagy (mitochondrial dysfunction and autophagy) by ameliorating glucose intolerance and insulin sensitivity (deregulated nutrient sensing) and by reducing telomere shortening (telomere attrition) and circulating pro-inflammatory cytokines (altered cellular communication/inflammaging).
NMN exerts these functions by enhancing metabolic reactions and enzymatic activities of sirtuins and PARPs [6] [7].
With aging, the entire body is affected. In this context, we would expect an anti-aging intervention to benefit many tissues in the body in contrast to the single-disease approach typically taken by the pharmaceutical industry. Which tissues and organ systems have been shown to benefit from NMN supplementation?
AG: Yes, you’re correct to note that our approach is not a single-pathology one. Indeed, due to NAD+’s involvement in physiologic processes like DNA repair, metabolism, and cell death, supplementation with NMN presents a high-potential therapeutic opportunity to address multiple age-related pathological processes.
In terms of organs, NMN administration has been demonstrated to have beneficial effects in several organs, including skeletal muscle, kidney, liver, heart, eyes, brain, pancreas, adipose tissue, and the vascular system [8].
In several pathophysiological conditions, NMN has shown to improve multi-organ insulin sensitivity and glucose tolerance, physical endurance, age- and diet-associated body weight gain, cardiac functions, large elastic artery stiffness, and capillary density.
It has also shown to protect from photoreceptor and corneal nerve fiber degeneration, renal and cardiac ischemia-reperfusion injury, and cognitive and memory loss.
The body of scientific literature backing the benefits of NMN is certainly impressive. Elevant Prime isn’t the only NMN supplement on the block, however. Could you talk a little bit about what differentiates your product from others?
GB: Rather than rely on third-party sources for the NMN in Prime (as well as other upcoming Elevant products), we developed our own proprietary manufacturing process and undertook extensive preclinical and clinical research on NMN itself.
This high degree of transparency is a key advantage of Prime over other NMN supplements. As I mentioned before, the online nature of the cellular optimization industry means that transparency and product accountability can be hard to find. In contrast, our proprietary formulation (NMN-C) is verified safe and is demonstrated to be well tolerated. A recent OECD 408 toxicology study – the first of its kind to be undertaken on NMN (based on our review of publications on PubMed as of January 1, 2021) – analyzed the No-Observable Adverse Effect Level (NOAEL) of NMN, with results showing our NMN-C to be an extremely well-tolerated option in comparison to other popular NAD+ boosters.
Furthermore, NMN-C acquired self-affirmed Generally Recognized as Safe (GRAS) status in accordance with stringent U.S. FDA regulatory guidelines. Performed by an independent panel of toxicology and nutrition experts, the successful self-GRAS review is a major endorsement of the quality and integrity of our NMN.
The proprietary manufacturing and formulation processes we use to create Elevant Prime were developed over several years in collaboration with leading NAD+ experts like Dr. Grozio. The result is a manufacturing process at cGMP-certified facilities in Europe that can be applied at a large enough scale to make safe, high quality NMN affordable for consumers.
We maintain strict control over every element of our processes, from production and manufacturing to dispatch, which ensures that Prime delivers what we are confident are the highest levels of quality available in an NMN supplement today.
The data backing up the safety of NMN-C is really robust which is crucial to any supplement. However, when we discussed the benefits of NMN, I’m guessing that most of those studies were conducted with other forms of NMN since NMN-C is a new product. Is there any reason to think that the efficacy of NMN-C may be different than other forms of NMN, for better or for worse?
AG: Our NMN-C formulation contains the same NMN molecule that is present naturally in our body. It will behave in the same way as the NMN used in studies that have demonstrated that NMN can provide myriad benefits as a result of boosting NAD+. It is the same molecule, so it will undergo the same biochemical reactions in the body.
One of the very positive things we are seeing specifically with NMN-C is that it is very well tolerated. The toxicology study I mentioned before determined a No-Observable Adverse Effect Level (NOAEL) for NMN-C to be = 1500 mg/kg/d, meaning that it is an extremely well-tolerated option. No other NMN has been tested in an OECD408 study, which means that the impurities coming with other NMN has not been proven to be safe at any dose. If you are curious, check out the full details of the study
We should also note that most of this efficacy data is from preclinical models such as mice and that translating these results into humans is notoriously difficult. What efficacy data for NMN in humans do we have so far? Do you know of any human studies in progress we should be keeping an eye on?
AG: Several human clinical trials are ongoing to assess the efficacy of NAD+ boosting strategies, of which NMN is a key one, in different pathophysiological conditions.
Given that the first clinical trial to evaluate the safety and bioavailability of NMN was only published in 2019 (single oral administration of NMN up to 500 mg was safe and effectively metabolized in healthy subjects without causing severe adverse effects), we are about to see a rapid increase in the body of clinical research being published for NMN.
In fact, just recently in April 2021, a human clinical study was published and showed that NMN improves metabolism in aged women with prediabetes [9]. Oral administration of NMN (250 mg/day) for 10 weeks showed significant improvement of skeletal muscle insulin signaling, insulin sensitivity, an increase of NAD+ levels in peripheral blood mononuclear cells (which give selective responses to the immune system and are the major cells in human immunity), and molecules derived from NAD catabolism/usage in skeletal muscle in postmenopausal obese women with prediabetes. Interestingly, NR administration (1 or 2g/day for 6 or 12 weeks) was not shown to improve insulin sensitivity in obese humans in several clinical trials [10] [11] [12] [13].
GB: Following soon will be the first results from our program of clinical research, which is assessing the kinetics and tolerance of NMN in human subjects. Two trials have already been completed on skin health and skin anti-aging, and two more will be completed by the end of June in Canada and France.
Five further trials are under preparation and will take place Q3 and Q4 2021.
These trials will assess the effect of NMN on different conditions, such as muscle recovery, metabolic syndrome, infection, and neurodegenerative disease. We have some of the world’s leading NAD+ and NMN minds undertaking these studies, which will help us continue to expand our platform of products that are evidence-based and grounded in scientific rigor.
Given that NMN has demonstrated efficacy and benefits in animal models of human diseases, we are really excited about several other clinical trials that we are aware of that are currently studying NMN’s clinical applicability. As efficacy data accumulates, awareness of scientifically validated consumer solutions will grow, meaning that more people will experience the potential benefits of NMN to health and lifespan sooner.
Thank you so much for taking the time to talk with us. We are definitely excited about the work that Elevant and its parent company Seneque are doing. Let’s end on future plans – can you give us a sneak peek at what is next for you two and the company?
GB: Thank you for the opportunity. It’s a very exciting time to be operating in the NMN and NAD+ boosting space. As Dr. Grozio mentioned before, in the very near future, we are going to be seeing more and more clinical studies published on NMN – not only from Elevant and Seneque, but also from other respected research organizations and sources within the cellular optimization industry. And – I don’t want to pre-empt their results, but we can’t ignore the potential benefits of these molecules already highlighted by existing research – we are confident that many of these findings are going to hold very positive indications for human health and longevity. We firmly believe that NMN-based interventions have the potential to spark a new era of therapeutic approach to health.
In Q4 2021, Elevant will launch a new patented formulation called Prime plus, which will contain NMN and a synergetic molecule drastically increasing its efficacy.
AG: I have always been fascinated by the pivotal role that NAD+ plays in maintaining homeostasis in our body and the plethora of cellular processes in which it is involved. So personally, I’m really excited to keep innovating with Elevant and Seneque by applying the latest technologies to refine our understanding on the biological processes in which NMN and NAD+ are involved. And, of course, to translate our discoveries into realities!
We would like to ask you a small favor. We are a non-profit foundation, and unlike some other organizations, we have no shareholders and no products to sell you. All our news and educational content is free for everyone to read, but it does mean that we rely on the help of people like you. Every contribution, no matter if it’s big or small, supports independent journalism and sustains our future.
Literature
[1] Saldeen, J., Tillmar, L., Karlsson, E. et al. Nicotinamide- and caspase-mediated inhibition of poly(ADP-ribose) polymerase are associated with p53-independent cell cycle (G2) arrest and apoptosis. Mol Cell Biochem 243, 113–122 (2003). https://doi.org/10.1023/A:1021651811345
[2] Avalos, J., Bever, K., Wolberger, C., Mechanism of Sirtuin Inhibition by Nicotinamide: Altering the NAD+ Cosubstrate Specificity of a Sir2 Enzyme. Molecular Cell, 17 (6), 855–868 (2005). https://doi.org/10.1016/j.molcel.2005.02.022
[3] Hwang ES, Song SB. Possible Adverse Effects of High-Dose Nicotinamide: Mechanisms and Safety Assessment. Biomolecules. 2020 Apr 29;10(5):687. https://doi.org/10.3390/biom10050687
[4] Ratajczak J, Joffraud M, Trammell SA, Ras R, Canela N, Boutant M, Kulkarni SS, Rodrigues M, Redpath P, Migaud ME, Auwerx J, Yanes O, Brenner C, Cantó C. NRK1 controls nicotinamide mononucleotide and nicotinamide riboside metabolism in mammalian cells. Nat Commun. 2016 Oct 11;7:13103. https://doi.org/10.1038/ncomms13103
[5] Giroud-Gerbetant J, Joffraud M, Giner MP, Cercillieux A, Bartova S, Makarov MV, Zapata-Pérez R, Sánchez-García JL, Houtkooper RH, Migaud ME, Moco S, Canto C. A reduced form of nicotinamide riboside defines a new path for NAD+ biosynthesis and acts as an orally bioavailable NAD+ precursor. Mol Metab. 2019 Dec;30:192-202. https://doi.org/10.1016/j.molmet.2019.09.013. Epub 2019 Oct 3.
[6] Fang EF, Lautrup S, Hou Y, Demarest TG, Croteau DL, Mattson MP, Bohr VA. NAD+ in Aging: Molecular Mechanisms and Translational Implications. Trends Mol Med. 2017 Oct;23(10):899-916. https://doi.org/10.1016/j.molmed.2017.08.001. Epub 2017 Sep 9.
[7] Covarrubias AJ, Kale A, Perrone R, Lopez-Dominguez JA, Pisco AO, Kasler HG, Schmidt MS, Heckenbach I, Kwok R, Wiley CD, Wong HS, Gibbs E, Iyer SS, Basisty N, Wu Q, Kim IJ, Silva E, Vitangcol K, Shin KO, Lee YM, Riley R, Ben-Sahra I, Ott M, Schilling B, Scheibye-Knudsen M, Ishihara K, Quake SR, Newman J, Brenner C, Campisi J, Verdin E. Senescent cells promote tissue NAD+ decline during ageing via the activation of CD38+ macrophages. Nat Metab. 2020 Nov;2(11):1265-1283. https://doi.org/10.1038/s42255-020-00305-3. Epub 2020 Nov 16. Erratum in: Nat Metab. 2021 Jan;3(1):120-121.
[8] Hong W, Mo F, Zhang Z, Huang M, Wei X. Nicotinamide Mononucleotide: A Promising Molecule for Therapy of Diverse Diseases by Targeting NAD+ Metabolism. Front Cell Dev Biol. 2020 Apr 28;8:246. https://doi.org/10.3389/fcell.2020.00246. PMID: 32411700; PMCID: PMC7198709.
[9] Yoshino M, Yoshino J, Kayser BD, Patti G, Franczyk MP, Mills KF, Sindelar M, Pietka T, Patterson BW, Imai SI, Klein S. Nicotinamide mononucleotide increases muscle insulin sensitivity in prediabetic women. Science. 2021 Apr 22:eabe9985. https://doi.org/10.1126/science.abe9985. Epub ahead of print.
[10] Dollerup OL, Christensen B, Svart M, Schmidt MS, Sulek K, Ringgaard S, Stødkilde-Jørgensen H, Møller N, Brenner C, Treebak JT, Jessen N. A randomized placebo-controlled clinical trial of nicotinamide riboside in obese men: safety, insulin-sensitivity, and lipid-mobilizing effects. Am J Clin Nutr. 2018 Aug 1;108(2):343-353. https://doi.org/10.1093/ajcn/nqy132.
[11] Dollerup OL, Chubanava S, Agerholm M, Søndergård SD, Altintas A, Møller AB, Høyer KF, Ringgaard S, Stødkilde-Jørgensen H, Lavery GG, Barrès R, Larsen S, Prats C, Jessen N, Treebak JT. Nicotinamide riboside does not alter mitochondrial respiration, content or morphology in skeletal muscle from obese and insulin-resistant men. J Physiol. 2020 Feb;598(4):731-754. https://doi.org/10.1113/JP278752. Epub 2019 Dec 26.
[12] Remie CME, Roumans KHM, Moonen MPB, Connell NJ, Havekes B, Mevenkamp J, Lindeboom L, de Wit VHW, van de Weijer T, Aarts SABM, Lutgens E, Schomakers BV, Elfrink HL, Zapata-Pérez R, Houtkooper RH, Auwerx J, Hoeks J, Schrauwen-Hinderling VB, Phielix E, Schrauwen P. Nicotinamide riboside supplementation alters body composition and skeletal muscle acetylcarnitine concentrations in healthy obese humans. Am J Clin Nutr. 2020 Aug 1;112(2):413-426. https://doi.org/10.1093/ajcn/nqaa072.
[13] Elhassan YS, Kluckova K, Fletcher RS, Schmidt MS, Garten A, Doig CL, Cartwright DM, Oakey L, Burley CV, Jenkinson N, Wilson M, Lucas SJE, Akerman I, Seabright A, Lai YC, Tennant DA, Nightingale P, Wallis GA, Manolopoulos KN, Brenner C, Philp A, Lavery GG. Nicotinamide Riboside Augments the Aged Human Skeletal Muscle NAD+ Metabolome and Induces Transcriptomic and Anti-inflammatory Signatures. Cell Rep. 2019 Aug 13;28(7):1717-1728.e6. https://doi.org/10.1016/j.celrep.2019.07.043.