Junevity Launches to Develop Cell Reset Therapeutics
Junevity, a biotechnology company on a mission to extend lifespan and healthspan by resetting cell damage from age-related diseases, today announced $10 million in seed funding led by Goldcrest Capital and Godfrey Capital.
The Junevity RESET platform is based on exclusively licensed research by co-founder Dr. Janine Sengstack at the University of California at San Francisco. RESET uses large-scale human data and AI to identify genes – or transcription factors – that can regulate cell damage. Then, it develops siRNA therapeutics against these targets to return cells to health. Junevity will use this seed funding to enhance the RESET platform and develop its first therapeutic candidates in Type 2 diabetes, obesity and frailty.
“My research at UCSF showed the power of targeting transcription factors to restore aged human cells back to health,” said Janine Sengstack, Ph.D., co-founder and Chief Scientific Officer at Junevity. “Based on these discoveries, we are bringing forward a new class of cell reset therapeutics for diseases, with the ultimate goal of greater human longevity.”
Diseases like obesity, diabetes, frailty, neurodegeneration and many others shorten human lifespan and are associated with complex cell damage at the transcriptional level. RESET uses billions of data points from human diseases and AI to rank and evaluate potential targets. Together, the platform outputs the Cell RESET Atlas, a collection of promising transcription factor targets by cell type and by disease for therapeutic targeting. Junevity then develops novel silencing RNA (siRNA) therapeutics to restore cellular transcription back to a healthy state.
Junevity’s preclinical data demonstrates the power of the RESET platform. In Type 2 diabetes, Junevity’s first siRNA therapeutic candidate improved glucose control and insulin sensitivity in diabetic mice without causing weight gain or other side effects associated with insulin sensitizers. In obesity, Junevity’s second siRNA candidate improved adipose tissue metabolism and reduced food intake, leading to 30% weight loss versus controls. Importantly, this weight loss was driven by fat loss with retention of lean mass. Both drug candidates are siRNA, meaning dosing once every 3-12 months is possible. This approach is patient-friendly and could increase compliance and satisfaction for diabetes and obesity treatments.
“Junevity’s RESET platform is a big idea that could broadly impact human health by addressing aging at the cellular level,” said John Hoekman, Ph.D., co-founder and Chief Executive Officer at Junevity. “We plan to advance multiple clinical programs, both directly and with partners, to make progress against diseases of aging.”
Junevity’s team includes world-class operators and advisors driven to extend human longevity, with an “outlier culture”based on mission, excellence, teamwork and intensity/pace. Junevity’s founding executive team includes:
- Dr. John Hoekman, Ph.D. – Co-founder, CEO – Created the technology for Impel Pharmaceuticals’ Trudhesa® nasal spray during his Ph.D. and led it to FDA approval in 2021
- Dr. Janine Sengstack, Ph.D. – Co-founder, CSO – Inventor of the RESET platform during her Ph.D. in Cellular Aging at UCSF
- Rob Cahill – Co-founder, COO – Previously machine learning researcher at UCSF and co-founder and CEO at Jhana, which was acquired by FranklinCovey (NYSE: FC)
“The Junevity team has a novel approach, incredible early data and tremendous potential to treat metabolic and age-related diseases,” said Brent Saunders, CEO and chairman of Bausch + Lomb, and an advisor to Junevity. “I’m excited to see how Junevity will advance this innovative platform.”
Junevity has exclusively licensed relevant technology from UCSF through its Office of Technology Management and Advancement. Junevity has since filed multiple composition-of-matter patents for its siRNA therapeutic candidates.
About Junevity
Junevity is a biotechnology company developing cell reset therapeutics for longevity. The Junevity RESET platform is the first to use large-scale human data and AI to identify transcription factor targets and repress them with siRNA therapeutics. The company is creating siRNA therapeutics to address diseases collectively impacting billions of people worldwide, including Type 2 diabetes, obesity, frailty and more. Based in San Francisco and founded out of UCSF in 2023, Junevity’s mission is to bring cell reset therapeutics to the world for longer lifespan and healthspan. Learn more at junevity.com.




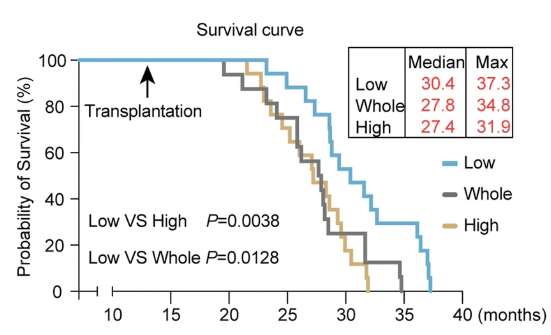



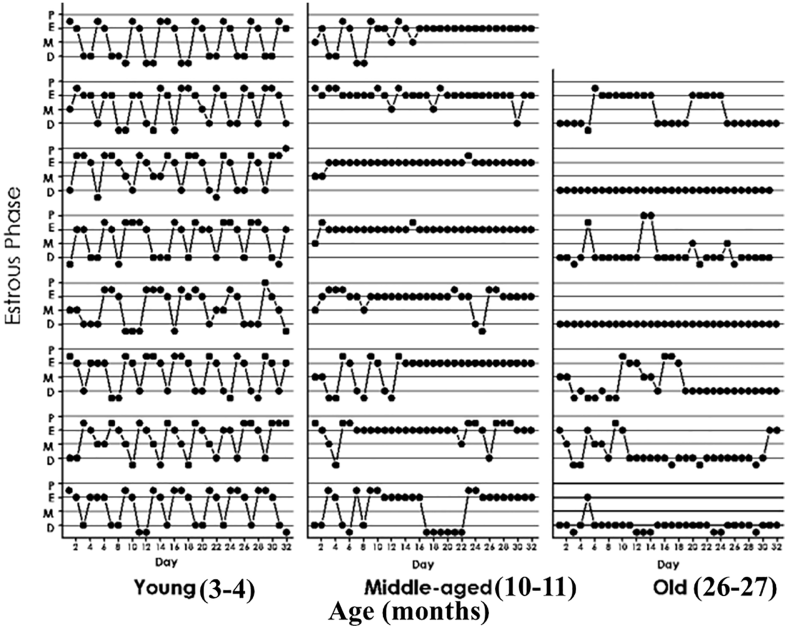
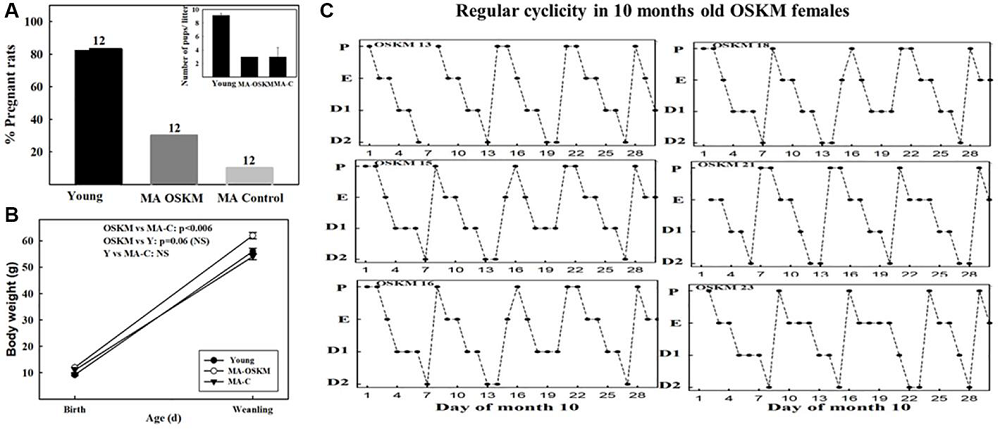

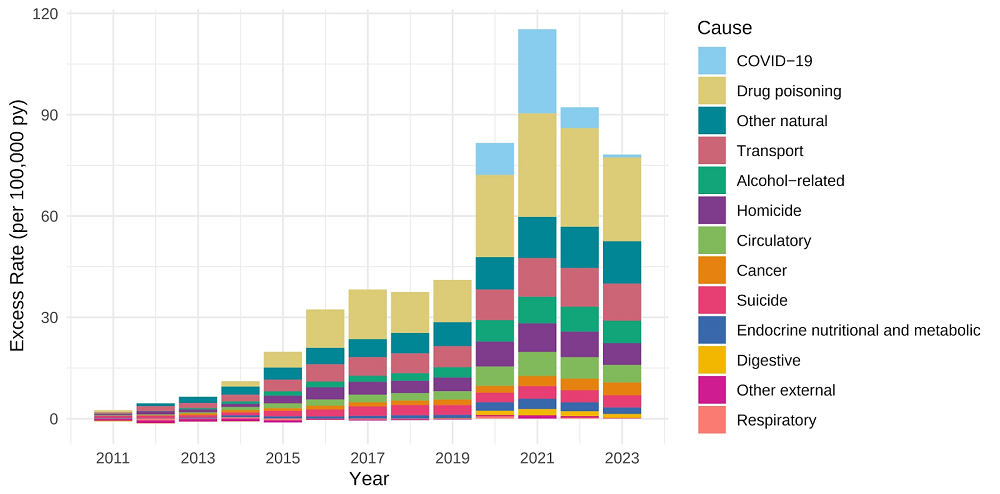


 nst disease and the persistent ravages of aging?
nst disease and the persistent ravages of aging?