BioAge Labs Announces Multi-Year Collaboration with Novartis
BioAge Labs, Inc. (“BioAge”), a clinical-stage biopharmaceutical company developing therapeutic product candidates for metabolic diseases by targeting the biology of human aging, today announced a multi-year research collaboration with Novartis. The collaboration aims to identify and validate multiple novel therapeutic drug targets by investigating the biological mechanisms that drive diseases related to aging and mediate the beneficial effects of physical exercise.
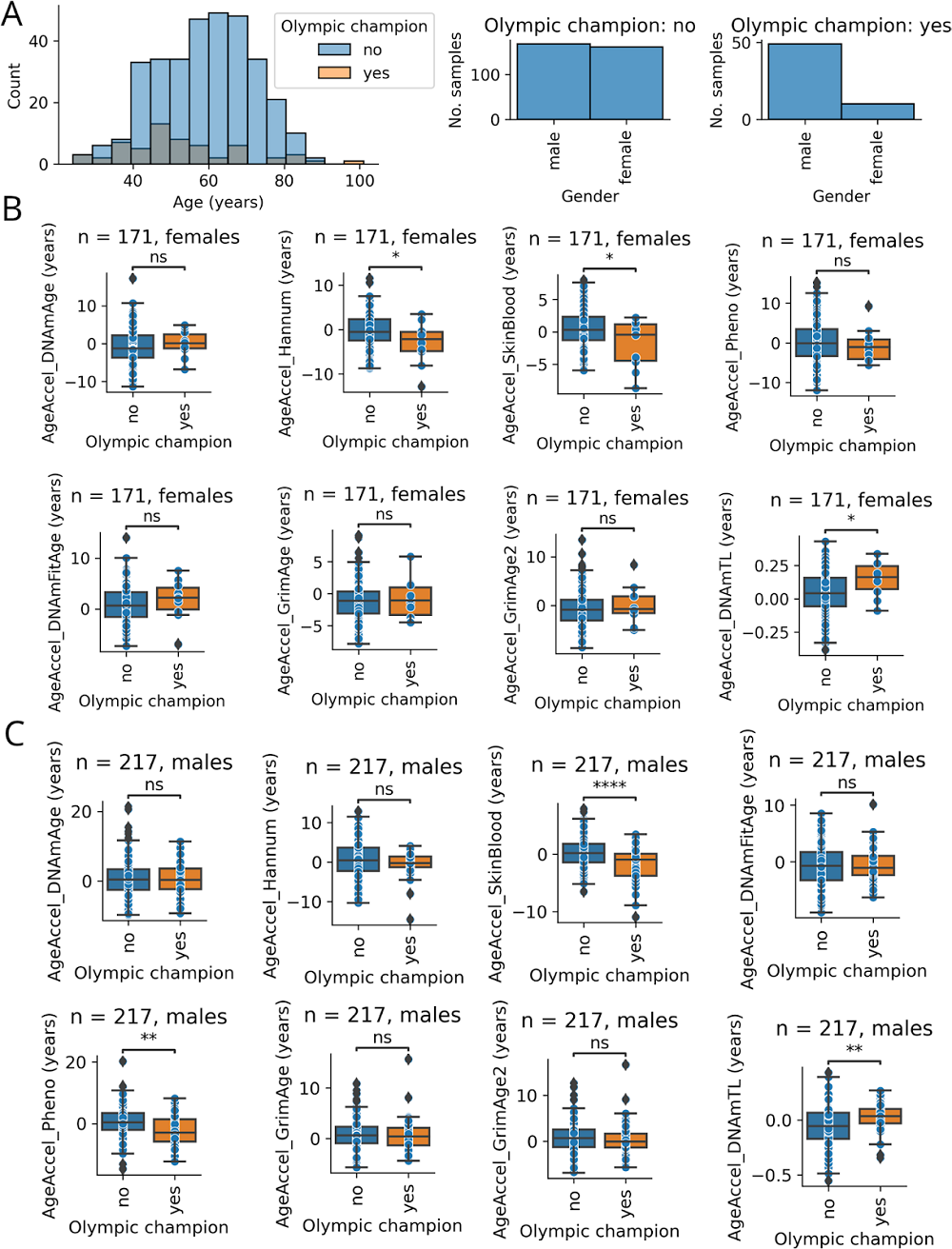
“Our platform, built on extensive longitudinal human longevity data, has allowed us to identify promising therapeutic pathways with significant potential to improve health outcomes,” said Kristen Fortney, CEO and co-founder of BioAge. “This collaboration with Novartis showcases the value of our platform and expands our capacity to discover and develop novel targets based on the insights from our data.”
The collaboration will leverage BioAge’s extensive proprietary human longevity datasets and Novartis expertise in exercise biology. BioAge’s proprietary discovery platform is based on exclusive access to longitudinal human aging cohorts followed for up to 50 years, combining detailed health records and functional measurements. Applying advanced analytics and machine learning techniques to this rich dataset enables BioAge to identify determinants of healthy lifespan, providing an engine for therapeutic discovery and development.
“We are excited to collaborate with BioAge, applying their human longevity data together with our scientific expertise in the biology of physical exercise to discover novel therapeutic targets,” said Michaela Kneissel, Global Head of Diseases of Aging and Regenerative medicine (DARe) at Novartis Biomedical Research. “By exploring the intersection of human aging biology and the biological drivers of the beneficial effect of physical exercise, we aim to bring forward novel treatment options for diseases related to aging.”
“The collaboration between Novartis and BioAge underscores the growing recognition that unraveling the biology of aging is a powerful approach to treating disease,” said Peng Leong, PhD, MBA, CBO and Head of Brain Aging at BioAge. “This collaboration represents a significant opportunity to accelerate our development of a broad portfolio of transformative therapies targeting novel mechanisms identified by our platform, dramatically expanding our therapeutic reach and benefiting patients across multiple indications.”
Under the terms of the agreement, BioAge will receive upfront payments and research funding of up to $20 million, plus up to $530 million in future long-term research, development, and commercial milestones. Novartis and BioAge each have the right to advance novel targets discovered under the collaboration and are each eligible to receive reciprocal success milestones and tiered royalties.
About BioAge Labs, Inc.
BioAge is a clinical-stage biopharmaceutical company developing therapeutic product candidates for metabolic diseases by targeting the biology of human aging. BioAge’s lead product candidate, azelaprag, is an orally available small molecule agonist of APJ that was observed to promote metabolism and prevent muscle atrophy on bed rest in a Phase 1b clinical trial. BioAge is also developing orally available small molecule brain penetrant NLRP3 inhibitors for the treatment of diseases driven by neuroinflammation. BioAge’s preclinical programs, based on novel insights from the company’s discovery platform built on human longevity data, address key pathways in metabolic aging.
Forward-looking statements
Statements in this press release about future expectations, plans and prospects, as well as any other statements regarding matters that are not historical facts, may constitute “forward-looking statements.” These statements include, but are not limited to, statements relating to anticipated preclinical and clinical development activities, timing of announcements of clinical results, trial initiation, and regulatory filings, potential benefits of azelaprag and the Company’s other product candidates and platform, the potential and timing of future milestone payments under the agreement with Novartis, and potential market opportunities for azelaprag and BioAge’s other product candidates. The words “anticipate,” “believe,” “continue,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “potential,” “predict,” “project,” “should,” “target,” “will,” “would” and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. Actual results may differ materially from those indicated by such forward-looking statements as a result of various important factors, including: BioAge’s ability to advance its product candidates, the timing and results of preclinical and clinical trials, the Company’s ability to fund development activities and achieve development goals, the Company’s ability to protect intellectual property, the Company’s commercial collaborations with third parties, the potential impact of global business or macroeconomic conditions, and the sufficiency of BioAge’s cash, cash equivalents and investments to fund its operations, and other factors discussed under the heading “Risk Factors” section of documents BioAge files from time to time with the Securities and Exchange Commission. Any forward-looking statements contained in this press release are based on the current expectations of BioAge’s management team and speak only as of the date hereof, and BioAge specifically disclaims any obligation to update any forward-looking statement, whether as a result of new information, future events or otherwise.
Contacts
PR: Chris Patil, media@bioagelabs.com
IR: Elena Liapounova, ir@bioagelabs.com
Partnering: partnering@bioagelabs.com