EARD2022 is over, but the research and events continue. Here’s a summary of everything that’s happened in August.
LEAF News
We are hiring! We are currently looking for a full-time chief of staff, a full-time data-driven Senior Marketing Manager, a part-time Youtube sponsorship/partnership acquisition lead, a social media intern, a part-time grant writer, and volunteers to support various programs. If you are interested in learning more about any of these positions, please contact us with your resume and salary expectations.
Cells Return from Death: Cells, dead for an hour under warm conditions, have been revived. Questions about when life begins have been hot topics for awhile, but there is also debate about when life ends.
Rapamycin and Metformin: Rapamycin and metformin, two well-studied drugs in aging research, can be combined for synergistic effects in mice. Rapamycin and metformin are viewed by many as the two most promising anti-aging drugs, but now scientists have found that these drugs can work hand in hand.
Steve Horvath on the Present and Future of Epigenetic Clocks: Dr. Steve Horvath is the inventor of the epigenetic clock and, currently, principal investigator at Altos Labs. We talked about the recent developments in this immensely important field, including pan-mammalian clocks, two-species clocks, and single-cell clocks, along with the challenges the field faces.
 Prof. Albert-László Barabási on Network Medicine: Albert-László Barabási is the Robert Gray Dodge Professor of Network Science at Northeastern University, and he also holds an appointment in the Department of Medicine at Harvard Medical School. We talked about a revolutionary ‘network medicine’ approach that can greatly enhance our ability to understand biological processes and seek cures for disease.
Prof. Albert-László Barabási on Network Medicine: Albert-László Barabási is the Robert Gray Dodge Professor of Network Science at Northeastern University, and he also holds an appointment in the Department of Medicine at Harvard Medical School. We talked about a revolutionary ‘network medicine’ approach that can greatly enhance our ability to understand biological processes and seek cures for disease.
Martin O’Dea Talks About the Longevity Summit: We recently had the opportunity to speak to Martin O’Dea about a new longevity-focused event happening in Ireland’s capital city on September 18th-20th. Martin holds an MBS and is a business lecturer at Dublin Business School in Dublin, Ireland. He is also the author of Beyond the Subjectivity Trap.
 Dr. Aubrey de Grey Will Speak at the Longevity Summit Dublin: We recently caught up with Dr. Aubrey de Grey and talked to him about the upcoming Dublin Longevity Summit and how things are looking on the advocacy landscape.
Dr. Aubrey de Grey Will Speak at the Longevity Summit Dublin: We recently caught up with Dr. Aubrey de Grey and talked to him about the upcoming Dublin Longevity Summit and how things are looking on the advocacy landscape.
Rejuvenation Roundup Podcast
Ryan O’Shea of Future Grind hosts this month’s podcast, showcasing the events and research discussed here.
Journal Club
Old Plasma Dilution Reduces Human Biological Age: The Journal Club has returned to our Facebook page with your host, Dr. Oliver Medvedik. This month, we have investigated a paper, “Old plasma dilution reduces human biological age: a clinical study”, in which Irina Conboy and her team investigated the effects of therapeutic plasma exchange on aging in people.
Research Roundup
 Vitamin D Fails to Improve Bone Health in Mega-Study: A high-quality, randomized, controlled trial found no effect of vitamin D supplementation or blood levels on the incidence of fractures in an aging population.
Vitamin D Fails to Improve Bone Health in Mega-Study: A high-quality, randomized, controlled trial found no effect of vitamin D supplementation or blood levels on the incidence of fractures in an aging population.
Hesperetin Upregulates Metabolism and Longevity in Mice: Researchers publishing in Journal of Biomedical Science have concluded that hesperetin, a compound found in various herbs, improves longevity in mice by promoting the expression of the pro-longevity gene Cisd2.
 Caloric Restriction Improves Immune System Function: A new study published in Mechanisms of Aging and Development has shown that caloric restriction effectively restores T cell abundance in aged mice. Caloric restriction has become a well-known anti-aging intervention, as it can reverse several hallmarks of aging and extend lifespan in different animal models.
Caloric Restriction Improves Immune System Function: A new study published in Mechanisms of Aging and Development has shown that caloric restriction effectively restores T cell abundance in aged mice. Caloric restriction has become a well-known anti-aging intervention, as it can reverse several hallmarks of aging and extend lifespan in different animal models.
Ghrelin Is Associated with Worse Muscle Aging in Mice: A team of researchers publishing through Multidisciplinary Digital Publishing Institute has described an association between ghrelin and skeletal muscle aging in mice. Ghrelin is a peptide containing 28 amino acids. Its main function is to stimulate the appetite through receptors in the hypothalamus.
 Sauna Combined with Exercise Improves Cardiovascular Health: In a randomized, controlled trial, scientists have shown that sauna and exercise, when taken together, might have a synergistic, beneficial effect on cardiovascular health and cholesterol levels. Sauna bathing has been credited with many health benefits, predominantly for the cardiovascular system.
Sauna Combined with Exercise Improves Cardiovascular Health: In a randomized, controlled trial, scientists have shown that sauna and exercise, when taken together, might have a synergistic, beneficial effect on cardiovascular health and cholesterol levels. Sauna bathing has been credited with many health benefits, predominantly for the cardiovascular system.
Developing Nanobodies to Fight Parkinson’s Disease: A team of researchers publishing in Nature Communications has described nanobodies that can destroy the α-synuclein aggregates that characterize Lewy bodies, which are associated with dementia and Parkinson’s disease. Traditional antibody therapies, while promising in some studies, are too large to enter cells in order to affect the aggregates there.
 Scientists Move the Boundaries of Post-Mortem Recovery: Researchers have been able to achieve substantial recovery of cellular and organismal activity in pigs that had been dead for a full hour. Advances in resuscitation have already moved the boundaries of life and death, making it possible to revive a person several minutes after the heart stops beating.
Scientists Move the Boundaries of Post-Mortem Recovery: Researchers have been able to achieve substantial recovery of cellular and organismal activity in pigs that had been dead for a full hour. Advances in resuscitation have already moved the boundaries of life and death, making it possible to revive a person several minutes after the heart stops beating.
An In-Depth Review of Skin Aging Genes: In a new systematic review published in Scientific Reports, multiple genes driving skin aging were identified. The authors start by explaining the intrinsic (genetic and chronological) and extrinsic (environmental) factors that drive skin aging.
 Hypertension Is Associated with Brain Drainage Changes: Researchers publishing in Aging have found that enlarged perivascular spaces in the brain are correlated with vascular disorders. These spaces, which are part of the brain’s glymphatic system, allow for the drainage of potentially dangerous metabolites such as beta amyloid.
Hypertension Is Associated with Brain Drainage Changes: Researchers publishing in Aging have found that enlarged perivascular spaces in the brain are correlated with vascular disorders. These spaces, which are part of the brain’s glymphatic system, allow for the drainage of potentially dangerous metabolites such as beta amyloid.
Rapamycin-Loaded Microneedles Reverse Hair Loss in Mice: Scientists have successfully regrown hair in a mouse model of hair loss using custom-made plastic microneedles loaded with rapamycin and epigallocatechin gallate (EGCG), an active ingredient in green tea.
 Identifying Mitonuclear Genes for Longevity: Publishing in GeroScience, a team of researchers that included Nir Barzilai and Matt Kaeberlein examined genes that may affect both mitochondria and lifespan.
Identifying Mitonuclear Genes for Longevity: Publishing in GeroScience, a team of researchers that included Nir Barzilai and Matt Kaeberlein examined genes that may affect both mitochondria and lifespan.
Dietary Restrictions Do Not Help Cognitive Function in Mice: A new study published in Neurobiology of Aging has shown that neither caloric restriction nor intermittent fasting improve late-life cognition in genetically diverse mice, but the effect depends on genetic composition.
 Combining Senolytic Pathways Has Synergistic Effects: A team of researchers have explained in Aging how multiple compounds that target the BCL-2 protein family are considerably more effective against senescent cells than each compound by itself.
Combining Senolytic Pathways Has Synergistic Effects: A team of researchers have explained in Aging how multiple compounds that target the BCL-2 protein family are considerably more effective against senescent cells than each compound by itself.
New Synthetic Molecule Alleviates Alzheimer’s in Mice: Scientists have synthesized a molecule that alleviates Alzheimer’s in a mouse model by targeting inflammation. Two of the most prominent – and probably interconnected – symptoms of Alzheimer’s disease are the accumulation of amyloid beta (Aβ) and chronic neuroinflammation.
 The Relationship Between Stroke and Inflammation: Publishing in Aging, a team of Chinese researchers has provided evidence showing a relationship between systemic inflammation and prognosis after a stroke. As the researchers point out, strokes are the leading cause of death in China.
The Relationship Between Stroke and Inflammation: Publishing in Aging, a team of Chinese researchers has provided evidence showing a relationship between systemic inflammation and prognosis after a stroke. As the researchers point out, strokes are the leading cause of death in China.
Almost Half of Cancer Deaths Worldwide are Preventable: Researchers have shown that 44.4% of cancer deaths worldwide can be attributed to preventable risk factors, including behavioral and environmental ones. It is well known that many cancer cases occur due to behavioral and environmental and factors such as smoking and pollution, which makes them theoretically preventable.
Plasma Dilution Appears to Rejuvenate Humans: Published in GeroScience, a groundbreaking study from the renowned Conboy lab has confirmed that plasma dilution leads to systemic rejuvenation against multiple proteomic aspects of aging in human beings. This paper takes the view that much of aging is driven by systemic molecular excess of signaling molecules, antibodies, and toxins.
 Mitochondrial Drug Alleviates Atherosclerosis in Mice: Scientists have drastically improved various symptoms of atherosclerosis in mice by precisely targeting mitochondria with a plant-derived antioxidant. Atherosclerosis, the accumulation of plaques on arterial walls, is one of the deadliest age-related diseases.
Mitochondrial Drug Alleviates Atherosclerosis in Mice: Scientists have drastically improved various symptoms of atherosclerosis in mice by precisely targeting mitochondria with a plant-derived antioxidant. Atherosclerosis, the accumulation of plaques on arterial walls, is one of the deadliest age-related diseases.
Intravenous Stem Cells Alleviate Guinea Pig Osteoarthritis: Scientists have shown that intravenous delivery of mesenchymal stem cells, which has some advantages over the more conventional intra-articular injection, alleviates age-related osteoarthritis and decreases inflammation in guinea pigs. Osteoarthritis, a degenerative joint disease, is one of the most common causes of disability in old age.
 Glycans as Biomarkers of Aging: In a new review published in Clinica Chimica Acta, researchers from the University of Zagreb discuss immunoglobulin G glycans, the changes that their composition undergoes with aging, and their potential as biomarkers of aging. One of the review’s co-authors is Prof. Gordan Lauc, who gave a presentation on them at EARD2022.
Glycans as Biomarkers of Aging: In a new review published in Clinica Chimica Acta, researchers from the University of Zagreb discuss immunoglobulin G glycans, the changes that their composition undergoes with aging, and their potential as biomarkers of aging. One of the review’s co-authors is Prof. Gordan Lauc, who gave a presentation on them at EARD2022.
A wearable electrochemical biosensor for the monitoring of metabolites and nutrients: The monitoring of metabolites for the early identification of abnormal health conditions could facilitate applications in precision nutrition.
Epigenome-wide association study analysis of calorie restriction in humans, CALERIE TM Trial analysis: DNA methylation changes may contribute to caloric restriction’s effects on aging.
Association of Leisure Time Physical Activity Types and Risks of All-Cause, Cardiovascular, and Cancer Mortality Among Older Adults: There were significant associations between participating in 7.5 to less than 15 MET hours per week of any activity and mortality risk.
Ginkgo biloba extract EGb 761 plus acetylcholinesterase inhibitors improved cognitive function in patients with mild cognitive impairment: These findings suggest that combined therapy with EGb 761 plus AChEI may provide added cognitive and functional benefits in patients with MCI.
Suppression of trimethylamine N-oxide with DMB mitigates vascular dysfunction, exercise intolerance, and frailty associated with a Western-style diet in mice: These therapies may be promising for mitigating the adverse effects of a Western diet on physiological function and thereby reducing the risk of chronic diseases.
Canagliflozin retards age-related lesions in heart, kidney, liver, and adrenal gland in genetically heterogenous male mice: Canagliflozin can be considered a drug that acts to slow aging and should be evaluated for potential protective effects against many other late-life conditions.
Fecal microbiota transplantation can improve cognition in patients with cognitive decline and Clostridioides difficile infection: This study revealed important interactions between the gut microbiome and cognitive function. Moreover, it suggested that FMT may effectively delay cognitive decline in patients with dementia.
Mitochondrial dynamics maintain muscle stem cell regenerative competence throughout adult life by regulating metabolism and mitophagy: As mitochondrial fission occurs less frequently in the satellite cells in older humans, these findings have implications for regeneration therapies in sarcopenia.
Long-lasting, dissociable improvements in working memory and long-term memory in older adults with repetitive neuromodulation: These findings demonstrate that the plasticity of the aging brain can be selectively and sustainably exploited using repetitive and highly focalized neuromodulation
Supplementing Glycine and N-Acetylcysteine (GlyNAC) in Older Adults Improves Aging Hallmarks: By combining the benefits of glycine, NAC and GSH, GlyNAC is an effective nutritional supplement that improves and reverses multiple age-associated abnormalities to promote health in aging humans.
News Nuggets
VitaDAO Funds ApoptoSENS Project for $253,000: Preventing the dysfunction of natural killer cells may be a promising area to explore in the fight against cellular senescence. Researchers are hoping to define the correlation between the increase in senescent cells and the onset or worsening of disease in humans.
VitaDAO Backs Research into Chronic Oral Disease: Periodontal disease affects more than 47% of adults aged 30 and over. For people over 65 years of age, that number rises to over 70%, making periodontitis one of the most commonly observed age-related illnesses. Jonathan An’s lab seeks to research inflammation-targeting compounds that can help treat periodontal disease.
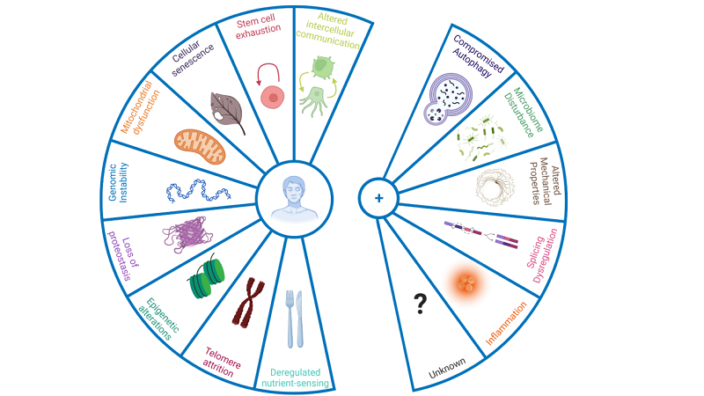
 Researchers Propose Five New Hallmarks of Aging: Publishing in Aging five months after their panel discussion in Copenhagen, many well-known researchers have explained their reasons for wishing to add new hallmarks of aging to the existing paradigm.
Researchers Propose Five New Hallmarks of Aging: Publishing in Aging five months after their panel discussion in Copenhagen, many well-known researchers have explained their reasons for wishing to add new hallmarks of aging to the existing paradigm.
Coming Up
SENS Research Foundation Announces Ending Aging Forum 2022: SENS Research Foundation has announced this year’s Ending Aging Forum, which will be held through a virtual conference platform with an immersive environment.
Longevity Investors Conference: Organized and sponsored by Maximon, the Longevity Investors Conference is focused on the investment aspects of longevity. The LIC welcomes everyone with an interest in the financial aspects of the longevity sector, including venture capitalists, asset managers, and managers of private equity funds and private banks.
Longevity Summit Dublin: This conference will feature two days of inspiring research developments along with top longevity entrepreneurs, biotech companies, longevity investors, and researchers from around the world.
We would like to ask you a small favor. We are a non-profit foundation, and unlike some other organizations, we have no shareholders and no products to sell you. All our news and educational content is free for everyone to read, but it does mean that we rely on the help of people like you. Every contribution, no matter if it’s big or small, supports independent journalism and sustains our future.