The annual Longevity Summit Dublin happened this June, and we are bringing you the highlights. Initiated by Aubrey de Grey and Martin O’Dea in 2022, this conference has earned a reputation for combining scientific depth with just the right amount of quirkiness over full four days of talks, panels, and late-night networking.
One of the Summit’s characteristic features is two awards: one for lifetime achievement and the other for a new star in the longevity field. lifespan.io executive director Stephanie Dainow was the latter’s first recipient back in 2022.
Unfortunately, we were only able to cover some of the several dozen talks at the conference. Our apologies customarily go to the equally worthy presenters who had to be left out.
Longevity goes mainstream
The first day began with a “pre-session” before the official opening, and the very first talk set an interesting tone. It was given by Michael Suk, the newly elected chair of the American Medical Association (AMA)’s Board of Trustees. The AMA is the biggest organization of medical doctors in the US, and having its leader talk at a longevity conference is an important event and an encouraging sign of our field inching closer to the mainstream.
Suk, through a remote connection, praised the recent scientific advances that bring us closer to personalized medicine that would allow for continuing healthspan extension. “The work you do here,” he said, “has the potential to change lives, to offer hope where there is none, and to redefine the future of healthcare. Together, we are building a future where technology and compassion go hand in hand, where surgical care is not just about precision, but about people, and where the promise of a longer, healthier life is within reach for all.”
Microsoft’s taking notice
Suk’s talk was followed by another high-profile speaker who does not come strictly from the longevity field: Sally Ann Frank, Worldwide Health and Life Sciences Lead at Microsoft for Startups and the author of The Startup Protocol: A Guide for Digital Health Startups.
Sally described Founders Hub, Microsoft’s platform for startup acceleration, which includes up to $150,000 in free Azure credits and other perks. She then moved on to explain how AI will revolutionize healthcare, which has become a recurring theme both in Dublin and at other longevity conferences.
Within Microsoft’s Startup Program, a smaller Pegasus Program is nestled to “curate a very small set of startups from key industries like health and life sciences.” With the Pegasus Program, Microsoft acts as a VC “to find those really pivotal, transformational solutions and accelerate their growth.”
Sally then gave an overview of Microsoft’s Pegasus portfolio, which includes companies such as Helfie.ai, the creator of an app that offers early diagnostics of various conditions using just a smartphone’s camera and microphone.
AI, Sally said, can help us understand aging at the molecular level for each person, predict future disease states, personalize treatments, develop new drugs and therapies, and, importantly, improve access to healthcare of underserved patients – something companies like Healfie.ai are already doing.
More on AI and longevity
The next speaker was another Microsoft alumnus, Tom Lawry, former National Director for AI – Health and Life Sciences at the company. Tom, too, is the author of a bestselling book: Hacking Healthcare. Today, he is managing director at Second Century Tech, an AI consultancy for health and medical leaders.
Tom started by drawing a line between predictive and generative AI. While, today, the latter is garnering all the attention with ChatGPT and the likes of it, the former has been used in healthcare for at least 12-15 years. Meanwhile, generative AI has seen yet to see massive use in healthcare, but its possibilities are endless.
While many novel technologies follow the so-called Gartner’s curve, where “the peak of inflated expectations” is followed by “the trough of disillusionment,” Tom thinks that AI will escape this fate and will just keep going strong, including in healthcare. One reason is that the amount of data is growing exponentially, with healthcare actually outperforming other fields. According to Tom, soon, 30% of all new data generated around the world will be related to healthcare.
At the same time, AI training costs are plummeting, which means that “doing interesting things with AI” is becoming easier and much more accessible. “By 2030,” Tom said, “it will probably be more expensive to take a friend out to lunch than to train a large data model.”
How will all this impact longevity? Tom highlighted AI’s ability to discover previously unknown correlations. For instance, deep learning AI was able to predict diabetes from a chest X-ray – the most ubiquitous type of health imaging available. However, in many situations, a mere smartphone camera can unlock amazing diagnostic capabilities. In this regard, Tom mentioned AEYE Health – the first company to receive FDA approval for diagnosing diabetic retinopathy, a leading cause of blindness, from handheld camera images.
AI will enable us to optimize health at all stages of life, instead of diagnosing diseases that have already developed, usually at an older age. Tom mentioned research that suggests millennials might be less healthy than previous generations (although that’s debatable), which makes early diagnostics and personalized proactive healthcare ever more important.
The founders’ words
The pre-session was followed by the official opening and two brief talks by the organizers – Aubrey de Grey and Martin O’Dea. In his talk, Martin made a case for nonprofits in the longevity field, such as the Longevity Escape Velocity Foundation (LEVF) that he co-founded with Aubrey.
LEVF’s flagship project is the Robust Mouse Rejuvenation Project (RMR), which tests combination therapies in mice. “What’s interesting about that project aside from the science,” Martin said, “is that nobody else would do it, even though a child can understand its value.”
Pharma companies cannot go in that direction, because their modus operandi is to look for a single novel intervention against a single indication. This leaves the important task of looking for possible life-extending combinations of existing therapies to non-profits.
The nonprofit activity in the longevity field must be greatly expanded, Martin argued, and companies that are profiting from the fundamental research performed years ago by noncommercial agents should be giving back to nonprofits.
The overarching goal, however, is to win over the public sentiment, which, Martin said, requires speaking more boldly about the longevity movement’s ultimate goal: “I think we need to say more in a kind of a confident, brave way that it’s the deterioration, it’s the aging that we want to tackle.”
Speaking next, Aubrey welcomed the participants and expanded on longevity escape velocity, the concept he has been promoting for decades that gave the name to his foundation.
LEV means buying time by eliminating age-related damage (another concept that Aubrey pioneered and is now entering the mainstream): “If you take someone who is, let’s say, 60 years both chronologically and biologically, and you rejuvenate them reasonably well so that they’re back to being biologically 40, then they won’t be biologically 60 again until they’re 80.”
However, some types of damage are harder to tackle, and they will continue to accumulate, but their effect will not be as devastating as today’s combined effects of all types of damage.
Hopefully, during those 20 extra years of life, geroscientists “will have been continuing to improve this rejuvenation arsenal,” Aubrey said. “We will be able to continue rejuvenating people because we will have improved the therapy so that some of the damage that used to be difficult to remove is now within the scope of the portfolio that we have. The idea is that there’s some finite, in fact quite modest, minimum rate at which we need people like the people in this room to be improving the comprehensiveness of rejuvenation technologies in order to stay one step ahead of the problem.”
For this to work, the longevity field must cast a wide net, bringing together people from various fields, which is what Longevity Summit Dublin is trying to achieve.
A task against AGEs
Revel Pharmaceuticals is one of those brave small companies to literally boldly go where no one has gone before: in this case, towards reversing the molecular damage produced by advanced glycation end products (AGEs). Like their name suggests, AGEs accumulate with age in blood and tissues, causing inflammation and the stiffening of the extracellular matrix by binding to molecules like collagen.
Aaron Cravens, Revel’s CEO, told the audience that his company is after a specific AGE, carboxymethylysine (CML). It exists in two forms: a free form present in cells and a bound form that accumulates in the extracellular matrix (for instance, in arterial walls). “The body has no physiological means of removing this,” Aaron said.
Revel built an enzyme engineering platform to develop bespoke enzymes. Currently, it focuses on removing the free-floating form of CML. According to Aaron, it strongly contributes to inflammation, in particular by binding to the RAGE receptor and initiating inflammatory signaling. The specific clinical application chosen is rheumatoid arthritis, an inflammatory disease, but the company envisions “crossing over into longevity biotech.”
The free version of CML has been shown to contribute to microglial dysfunction in the brain, which, in turn, is linked to neurodegenerative diseases. “We’re developing enzymes that can specifically reverse these changes as they build up,” Aaron said. “It’s not trying to slow or prevent, it’s truly reversing the damage.”
The company has gone a long way, improving their enzymes’ ability to remove CML a million-fold. Aaron admitted that bound CML is a more hard-to-crack target, but Revel is making progress. In the future, they also hope to tackle even more insidious AGEs, such as glucosepane.
The oldest of the old
In a talk that was less about cutting-edge science but important nonetheless, Natalie Coles, the chief phlebotomist and project manager for R3Bio, described her work on creating a supercentenarian biobank.
Supercentenarians are a very rare breed. At any given time, there’s only a few hundred of them in the world. Their prevalence is growing, but not as fast as that of centenarians (whose population is booming), highlighting just how much harder it is to cross the 110-year line.
According to Coles, who has been studying supercentenarians for years, they are more biologically homogeneous than centenarians: meaning that they possess, to paraphrase a popular movie, “a very specific set of biological skills” that allows them to outlive the rest of us – and those features are worth studying in depth.
Coles engages with supercentenarians personally, travelling all over the world to take biological samples from them. Her talk focused on the intricacies of performing this job and on her interactions with supercentenarians.
The challenges begin with validating the age claim. One organization that does this is the Gerontology Research Group (GRG) (Wacław Kroczek from GRG also presented at the conference, detailing the company’s validation methodology). For people born at the beginning of the previous century, especially in underdeveloped parts of the world, proper documentation might be hard to find.
After the claim has been verified, Natalie arrives to collect samples and perform interviews. Everything has to be arranged as fast as possible, since the average remaining lifespan for a supercentenarian is just one year. Many of Natalie’s patients died before she could meet them.
Less than one tenth of supercentenarians are males, which makes them especially valuable for researchers. Natalie mentioned the robustness of supercentenarians’ immune systems, made evident by the COVID pandemic: most of the supercentenarians who had gotten infected survived the disease, and at least one person did it twice.

Geroscience is the key
Michael Ringel of the Boston Consulting Group talked about the recent trends in the longevity field. After decades of not taking geroscience seriously, there is apparently a growing understanding in society that investigating the biology of aging is the only way to ward off the economic threats of population aging and to meaningfully increase human lifespan.
If we do nothing, global resources might be devastated by the need to care for the increasingly aged population, exacerbated by slumping birth rates. However, even a slight slowing of the rate of aging would bring unprecedented prosperity, adding hundreds of trillions of dollars to the global economy over the course of a decade.
Today’s medicine is clearly unable to achieve this, as the gains in life expectancy for every additional dollar spent on healthcare have flattened out. Thankfully, Michael said, “we’ve seen significant growth in aging research, investment, and new company formation over the last couple of years.” Aging research is outperforming biomedical research in general and mentions of longevity in the media are also on the rise.
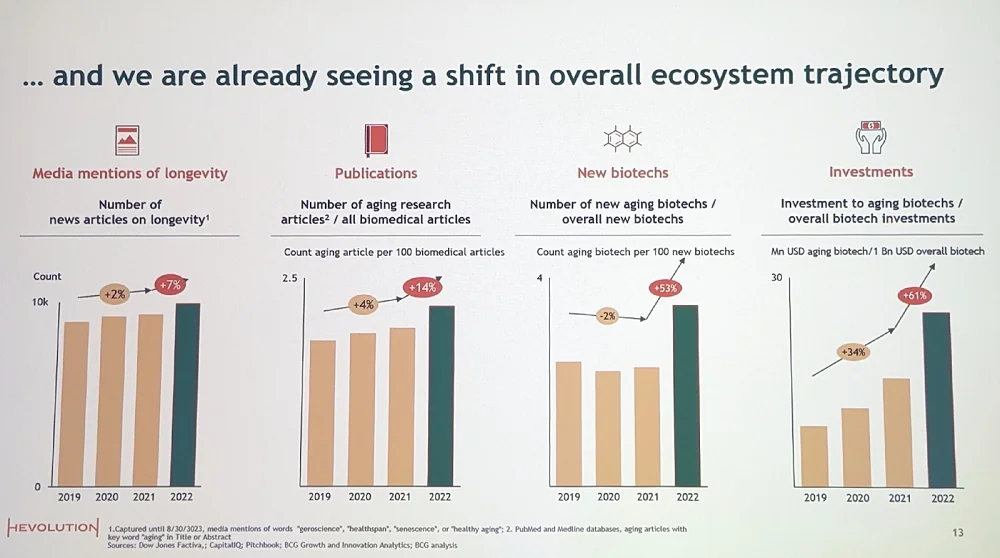
Importantly, Big Pharma is finally taking notice. Numerous pharmaceutical companies have partnered with longevity biotech startups working on therapies that are built on the principles of geroscience. Among the most notable collaborations are the ones between AbbVie and Google’s Calico, and between Pfizer and Gero. Answering a question from the audience about the seeming lack of output from Calico, which exists since 2013, Michael noted that AbbVie seems happy about the collaboration.
Senescence and mTOR
Lynne Cox from Oxford University is fascinated with cellular senescence. Senescence, she said, is very tightly connected to aging: “All of the things that drive aging also drive cellular senescence.” On top of that, senescence is itself a hallmark of aging, contributing to other hallmarks and age-related diseases. The senescent cell burden grows faster with premature aging, and injecting senescent cells into a healthy animal accelerates aging.
While senescent cells are heterogeneous, they also “show very marked changes in gene expression.” One particular change in which Lynne is very interested is the upregulation of the mechanistic target of rapamycin (mTOR). Lynne’s group has been working for years on an in vitro model – human skin fibroblasts – which enabled them to dig deep into the molecular aspects of senescence.
When a cell goes senescent, its mTOR pathways stop reacting to regular stimuli such as amino acid abundance. Instead, they are constantly switched on, which is intriguing since mTOR is associated with growth and senescent cells no longer divide. However, they grow much larger than non-senescent cells of the same type.
First generation mTOR inhibitors, such as rapamycin, mostly act on one of its parts, mTOR complex 1 (mTORC1). While some scientists are working on creating even more specific mTORC1 inhibitors, Lynne’s group has chosen the opposite direction: to target both mTORC1 and mTORC2. For this, they are using an investigational drug from Astra Zeneca, AZD8055, an ATP mimetic that competes with ATP for the active site of mTOR.
Chronic mTOR inhibition with this drug greatly increased fibroblasts’ replicative lifespan, even when administered intermittently to very aged cells. In already senescent cells, the treatment reversed various phenotypes of senescence. It alleviated age-related telomere loss, cytoskeleton abnormalities, impaired mitophagy, and morphological changes.
Lynne’s group found that senescent cells are very active mitochondria donors. While healthy cells also exchange mitochondria from time to time, senescent cells do it much more often, populating adjacent cells with their damaged mitochondria via “nanotubes”, which might be one of the mechanisms of spreading senescence across cell populations. This process was also significantly abrogated by the treatment.
Lynne and her colleagues are moving through with a clinical trial of rapamycin in older adults. It will ask questions such as whether rapamycin affects aging muscle strength and response to resistance exercise and whether it can protect the immune system from aging in vivo in humans.
CHIP: Attack of the clones
The Rising Star Award this year went to Alexander Fedintsev from the Radical Life Extension Group. In his talk, Fedintsev proposed a new hallmark of aging: clonal hematopoiesis (CHIP). Clonal hematopoiesis is a condition in which a single hematopoietic stem cell (HSC) acquires genetic mutations that give it a growth advantage, allowing it to expand clonally within the bone marrow. This results in a significant proportion of blood cells being derived from this single mutated stem cell rather than from a diverse population of HSCs.
Mutations that increase self-renewal also impair HSCs’ ability to produce healthy blood cells. Hence, in an evolutionary process, less immunocompetent but more rapidly reproducing cells soon outcompete normal cells, similar to how cancerous cells outcompete non-cancerous ones. CHIP is also associated with lymphoid/myeloid bias, in which HSCs preferentially differentiate into myeloid lineage cells (e.g., granulocytes, monocytes) rather than lymphoid lineage cells (e.g., B cells, T cells). Myeloid bias also has a detrimental effect on the immune system.
The prevalence of CHIP rises exponentially with age and causes a significant skew towards the innate immune response that myeloid cells are associated with. Innate immunity is highly pro-inflammatory, which might be one of the causes of age-related sterile inflammation (inflammaging). As happens a lot in biology, this creates a positive feedback loop, with inflammation promoting CHIP.
CHIP drastically increases the risk of diseases: of cardiovascular disease by 80%, of blood cancer by tenfold, and of lung cancer by 60%. Some research indicates that CHIP might be a leading cause of death in extremely old people. Clonal expansion also happens in other cell types, such as in mesenchymal stem cells, and might contribute to atherosclerosis.
Currently, we don’t have good tools to combat CHIP. However, prolonged inhibition of the pro-inflammatory cytokine IL-6 has shown some promise, including in non-human primates. Answering a question about how safe lifelong IL-6 inhibition is, Fedintsev noted that rheumatoid arthritis patients receive such therapy, which only causes a slight increase in infections but is associated with less CVD risk.

Alexander Fedintsev receives the award from Aubrey de Grey. Photo: lifespan.io
Peter Diamandis on XPRIZE Healthspan
Entrepreneur and philanthropist Peter Diamandis tuned in remotely to talk with Aubrey de Grey about longevity and specifically XPRIZE Healthspan. Peter is Executive Chairman of the Board of XPRIZE Foundation, which he launched 30 years ago. Across those three decades, 30 prizes have been launched in domains ranging from space exploration to biodiversity.
Peter and Aubrey reminisced on their first rounds of discussing the possibility of a longevity XPRIZE almost two decades ago. While Peter was already a longevity fan, he felt that the field was too small, and the time wasn’t right back then.
It changed several years ago, when the entrepreneur Sergey Young, Peter’s partner at BOLD Longevity Growth fund, entered the picture. Young, a passionate longevity investor, helped raise the first one million dollars to develop the ideas for and the design of the future prize. With the longevity field growing fast, the idea seemed much more plausible.
One problem was how to measure success. Eventually, it was decided to offer the prize to the first team that can reverse functional loss due to aging. The main focus is on the domains of cognition, muscle health, and the immune system. “Minimum is 10 years reversal,” Peter said, “but the goal is 20 years”. Aubrey de Grey joined the team as an advisor, and several benefactors, including Hevolution foundation, helped put together the unprecedented prize fund.
Before that, the largest XPRIZE ($100 million), funded by Elon Musk, was for developing carbon capture technologies to help fight climate change. However, given the immense importance of increasing healthspan for humankind, the organizers agreed that the new XPRIZE should symbolically be even bigger, and it was set at $101 million.
Musk’s name having been mentioned, Aubrey asked Peter, why doesn’t Musk support longevity? Peter replied that he was able to eventually make Musk more amenable to the idea of life extension: “Elon feels that science makes progress one death at a time, that people need to die in order to make room for new people. And I said to him, Elon, no one at GM had to die to create Tesla.”
Today, the giant prize is open for applications, and Peter called on scientific teams to register. He is confident that longevity is on track to become one of the biggest markets in the world. His other call was for billionaires (although there probably weren’t any in the audience) to use their money “to make a dent in the universe.”
Pros and cons of telomere extension
The Lifetime Achievement Award was given this year to Maria Blasco, a veteran geroscientist whose contribution to the longevity field is formidable. After a short ceremony Maria proceeded to give a keynote talk on telomeres, the leading topic of her research for the last three decades.
At the dawn of modern geroscience, telomeres captivated the young field’s attention. Many hopes were tied to them, and telomere attrition was included in the original nine Hallmarks of Aging (Maria was a co-author of the seminal Hallmarks paper from 2013).
It turned out that not everything revolves around telomeres, and their role in health and longevity is complex and multifaceted. For instance, many types of cancer cells turn telomerase expression back on to maintain the length of their telomeres, ensuring continuous proliferation.
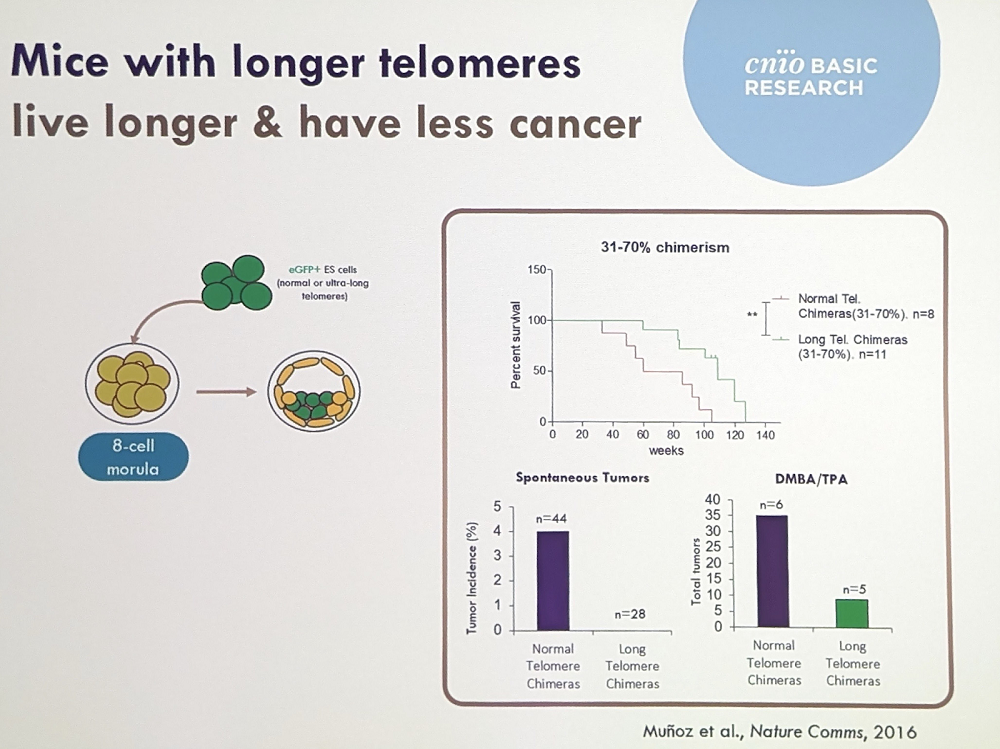
Maria described some of the most important experiments with telomeres. For instance, telomerase deficiency in mice causes accelerated aging, while transgenic mice with extra-long telomeres live longer and, importantly, get less cancer, which is the leading cause of death in lab mice. These mice also showed significant improvements in healthspan: better metabolism, less cognitive decline, less osteoporosis, and so on.
Telomere shortening plays an important role in many diseases, including the deadly pulmonary fibrosis. Gene therapy with TERT (telomerase reverse transcriptase, a crucial telomerase subunit) stops and even reverses pulmonary fibrosis in a mouse model, reducing inflammation, damage, and senescent cell burden.
Other conditions in which short telomeres are a factor include kidney fibrosis and myocardial infarction. In one study, only mice with short telomeres developed kidney fibrosis when challenged with folic acid.
Another study that Maria mentioned might provide an explanation of why turning back telomerase expression seems to lower the risk of cancer instead of elevating it: shorter telomeres cause chromosomal instability, which increases the risk of oncogenic mutations. Genetically engineered TERT-expressing mice were more protected from cancer even when challenged with oncogenic mutations.
A study from 2020 tied COVID severity to shorter telomeres. Another one, from 2024, detected more lung fibrosis in COVID survivors with shorter telomeres. Telomeres might not hold the ultimate key to life extension, but they certainly play an important role in aging and age-related diseases that needs to be thoroughly investigated.
Eat less, live longer
Rozalyn Anderson of the University of Wisconsin Madison reported on another longevity “staple”: caloric restriction (CR). CR, studied since the mid-20th century, has been shown to improve healthspan and lifespan in many model organisms. There are also some initial positive findings in humans. For instance, the CALERIE randomized controlled trial showed “a persistent and significant reduction of all measured conventional cardiometabolic risk factors” as a result of long-term 25% caloric restriction (although Rozalyn thinks that due to weak adherence, the actual CR rate in CALERIE was around 15%).
Caloric restriction affects many mechanisms of aging, and Rozalyn’s group is working on understanding how exactly this happens. “Metabolite, energy, and oxidation sensors regulate longevity and all have been implicated in CR’s mechanisms,” Rozalyn said. “Metabolism influences all cellular functions from information access to transport to movement and growth to signaling.” There is a metabolic component to cardiovascular disease, neurodegeneration, cancer, and diabetes, to name a few. Unsurprisingly, many geroprotective interventions work along the same pathways that CR does.
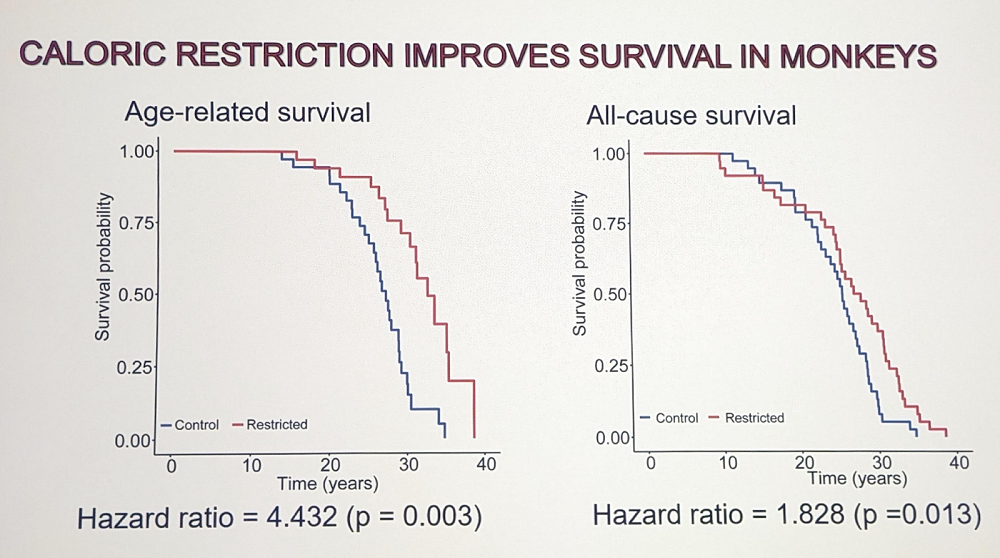
Rozalyn told the audience about the important and long-running study of CR in rhesus monkeys, which are long-lived and very humanlike animals. They share 93% of their genome with humans and have similar aging outcomes. 76 monkeys were recruited for this study in the late 80s / early 90s and monitored for more than 30 years, and now we have lifespan data.
Lifelong 30% caloric restriction had a profound effect on lifespan and survival, lowering the odds of dying from age-related conditions by 4.4 times. The scientists identified multi-omic CR signatures and recorded some intriguing effects. For instance, calorie-restricted animals had less muscle mass in midlife but more in late life: they were partially immune to age-related loss in both muscle quantity and quality. CR also countered the decline in voluntary physical activity that is characteristic of old age. Both males and females responded to CR, but somewhat differently.
Building a mega-movement
Melissa King has over two decades of experience in business, non-profits, and public affairs. About two years ago, she co-founded Healthspan Action Coalition with another veteran medical research advocate, Bernard Siegel. We excitedly reported on the creation of HAC from Longevity Summit Dublin 2022, welcoming those advocacy heavyweights into our field. Since then, the coalition has grown impressively to unite more than 160 organizations across 25 countries, including lifespan.io.
Melissa has steered important campaigns and projects in patient advocacy and biomedical research funding. For example, she led a successful ballot initiative in California to secure $8.5 billion in funding for the California Institute for Regenerative Medicine, which specializes in stem cell research.
Patient advocacy, Melissa argued, is impressively effective in mobilizing public opinion and funds for health research initiatives. Now is the time to apply the principles of patient advocacy to prolonging healthspan. After all, when it comes to aging, every person on the planet is a patient. This creates a potential for a truly massive movement.
While we are all excited about the rise of longevity biotech, “public funding for science is what brought us to space and the moon, enabled medical research that brought us vaccines and other life-saving treatments,” Melissa said. “Private industry and funding usually don’t come in early because of the risk.”
Government oversees immense resources, but it needs to be steered in the right direction by public opinion. While many at this and other recent conferences evoked working with public opinion as a crucial element of the longevity field’s success, Melissa made it the cornerstone of her talk.
Skillful and effective science communication is the key, she said, and we couldn’t agree more. Melissa called on the members of the geroscience community “to help the public by making it a point to communicate with non-scientist friends and family members about science and your work.” This can be done by writing articles/op-eds for lay audiences, giving talks, and more.

We would like to ask you a small favor. We are a non-profit foundation, and unlike some other organizations, we have no shareholders and no products to sell you. All our news and educational content is free for everyone to read, but it does mean that we rely on the help of people like you. Every contribution, no matter if it’s big or small, supports independent journalism and sustains our future.