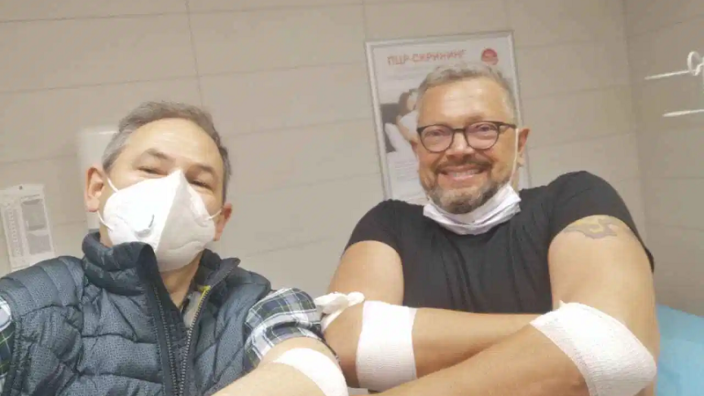
We interviewed a group of Russian biohackers who performed a plasma dilution experiment on themselves. This experiment, the first of its kind, was based on previous mouse studies by Drs. Irina and Michael Conboy.
Some molecules, while essential for various body functions, can be harmful when overproduced. Inflammatory cytokines, such as transforming growth factor beta 1 (TGF-ß1), interleukin 6 (IL6), and tumor necrosis factor alpha (TNFa) are good examples. The concentration of these cytokines in our blood rises with age, provoking inflammaging, the chronic inflammation that is associated with aging. It has been long speculated that reducing the harmful molecules in circulation can attenuate aging.
Back in 2005, Drs. Irina and Michael Conboy created a furor with their research on parabiosis, which links two vascular systems together. The Conboys connected the vascular systems of young and old mice and showed that as a result of the blood exchange, old mice became younger and vice versa [1]. This discovery spurred a flurry of research activity; for instance, earlier this year, we reported on some highly promising results of adding a cocktail of young blood factors to the bloodstreams of aging mice. However, the Conboys have always maintained that it is what we take out of the bloodstream that matters more. A few months ago, they showed that mere dilution of blood plasma with saline can produce a considerable rejuvenating effect (read our June interview with them). Later, in November, they published another paper that demonstrated restoration of cognitive functions following plasma dilution.
However, all this research has been done on mice, which led a small group of Russian biohackers to take matters in their own hands. Biohacking is a form of citizen science: do-it-yourself biology experiments. For biohackers focused on longevity, this includes performing experimental treatments (often on themselves) or repurposing existing treatments to improve health and, hopefully, wind back biological age.
The group’s scientific advisor, Alexander Fedintsev (read our interview with him) devised a protocol for plasma dilution in humans and a panel of biomarkers to watch. Then, following some logistical wizardry, the procedure was performed on two volunteers. Though not a scientific study per se, this experiment produced interesting, overall positive, results that can potentially influence and guide further research. Our interviewees think that biohacking, when done right, may become an important factor in the longevity field.
Alexander Fedintsev (scientific advisor)
How did your group first get interested in the idea of plasma dilution? I understand that Irina Conboy’s work had a certain influence?
Not just influence. It played a central role. The Conboys’ study was published in May. It showed that simple plasma dilution can recapitulate most of the benefits of parabiosis. The original parabiosis results hinted on the existence of certain systemic factors of aging and at the possibility of its reversal. This recent study made the procedure easier and eliminated ethical controversies. The procedure is almost similar to donating blood plasma. Only the liquid fraction is drawn. It does not contain blood cells, such as erythrocytes, leukocytes, and thrombocytes – just the liquid part with signaling molecules dissolved in it. So, the Conboys drew half of the plasma from their mice and replaced it with normal saline and some albumin. Albumin is an important transport protein, and they probably thought that extracting that much albumin from the bloodstream can be harmful, so they wanted to replenish it. This simple procedure yielded some interesting results: it triggered muscle regeneration in mice, liver regeneration in older animals, and improved neurogenesis. Recently, in late November, I think, another study was published that showed some real cognitive improvement following this procedure. So, now we have some serious proof that blood contains signaling molecules that harm the organism, but there is no data on whether this procedure actually prolongs lifespan. I think there is a reason for it. It is highly unlikely that this procedure results in any meaningful life extension. I think most of the effect is on healthspan rather than on lifespan. It is still good news, since we currently have very few ways to extend healthspan.
Why did you decide to participate in this small-scale experiment on humans?
Our team has existed for some time now. It is a small community of biohackers. It seemed like a great way to quickly test this intervention, get some results fast, and tell people about it.
What was your role?
I designed the experiment, developed the biomarker panel that we used, and worked on the logistics on how to make it all happen considering our modest means. We could not just follow the Conboys’ mouse study protocol, so we found a way to adapt it to a human experiment in order to do something very close to what the Conboys did.
What kind of problems did you encounter while working on the protocol?
The first problem that made us delay the experiment for six months was the pandemic. We could have done it sooner, the experiment being so simple. The second problem was that current medical protocols for plasma dilution in humans limit the amount of plasma that can be drawn, so it had to be done in several sittings. We had to calculate how many times we needed to draw plasma so that, in total, about half of the plasma would be replaced. Then, we had to figure out how to inject albumin. The medics who drew the plasma refused to do it, so albumin had to be injected immediately after the plasma donation by a different doctor. Then, there was the development of the biomarker panel – we had to figure out what to look at and at which day since the experiment.
How did you choose the tests for the panel?
It would have been interesting to look at cognitive and muscular markers, but both our participants were too young: 50-60 years old. They probably do not have sarcopenia or cognitive decline yet, so there was no way for us to measure it. We chose different biomarkers, such as liver function – both of our participants had had some abnormalities in their liver biomarkers. We wanted to check kidney function because it declines with age. We checked the immune system, because as we age, the number of naïve T cells declines, and these are indispensable for fighting new infections. Immunosenescence is a hot topic in times of COVID. Hematopoietic cell aging is characterized by a shift towards myeloid progenitors. We looked at the ratio of neutrophils and lymphocytes, how it changed. Cholesterol is another important marker in the lipid profile of blood. We did a very comprehensive lipid profile that included a rare biomarker that many labs do not check for – oxidized low-density lipoproteins (Ox-LDL). I can say that this marker plummeted all the way down to its normal level in one participant that had it elevated prior to the procedure. We also checked for various hormones, including insulin-like growth factor (IGF), that are related to aging and lifespan, and many other markers, including biochemical ones, such as urea and uric acid, along with oxidative stress markers, such as lipid peroxidation products and glutathione. Contrary to epigenetic clocks, these markers can be clinically interpreted.
Do you plan to publish the results, maybe as a case study?
We have all the data published as a Google spreadsheet on our website so that researchers can see it. We do not plan to publish an article. First, I am convinced that soon we will have full-scale clinical trials of this method, maybe by the Conboys, and there is something in the works here in Russia as well. I do not know how valuable our data is, considering our sample size was just two people. We just wanted to see whether it was possible to arrange such an intervention in humans using the means we had at our disposal, and whether it would do any good. Now we know it actually did some good, in terms of the number of naïve T-cells, levels of oxidized LDL. The drop in Ox-LDL levels was probably due not simply to dilution but to some deeper processes, because in one participant, these levels declined, while in the other they went up from an originally low level. So, in both participants, LDL levels normalized and stayed normal for at least two weeks. Liver markers improved by a lot, and the myelocyte/lymphocyte ratio improved. There were some controversial results, such as one participant having insulin levels decline four-fold but not the other one.
Seems like we are looking at an optimization of certain parameters rather than just up- or downregulation.
Yes, the shift sometimes happened in opposite directions. It contradicts the hypothesis that all this procedure does is plasma dilution. It is important to add that we included some inflammation markers, such as C-reactive protein and IL-6. Most of these markers went down in both participants, which points to a decline in systemic inflammation. These markers are very important from the standpoint of aging.
Which results were unexpected?
For instance, cholesterol went up in one of the volunteers. We expected it to go down in both participants because of the dilution. Probably, cholesterol levels can go back to normal faster than we thought. Also, the insulin levels. We also did not quite anticipate the 40% surge in the number of naïve T cells. The ratio between naïve T-cells and memory T-cells went up too – by as much as 30%.
Do you have an explanation for it?
Not yet, and I would wait for more data before hypothesizing. We only had two participants, it could be random.
Do you plan to expand the project? Maybe recruiting new participants?
We would like to have more people tested, but we do not plan to turn it into a large study. Let other people do it. We wanted to prove that the concept can be widely available and easily organized. What we would like to see is if albumin supplementation is essential. I suspect that it is not, at least in relatively young people. This would mean that the procedure can be simplified even further.
Do you worry about biohackers trying such serious procedures?
I think no one should be denied the right to experiment on themselves if they understand the risks and know what they are doing. Most of what is now being peddled as biohacking caters to people’s ignorance. True biohacking, on the other hand, is science, but it is less regulated than official science. If the studied effect is big, the study size can be smaller. Such experiments can be a part of science as well. I think it is important for research.
Anything else you would like to tell our readers?
I would ask them not to develop high expectations of this method, despite us having some good results with the markers. It still does not mean we will achieve life extension any time soon. Even if these markers are indeed causally linked to aging, we might not be able to achieve a lingering effect. It is possible that the efficacy of the procedure declines with age. Senolytics and plasma dilution may not work in very old people. One possible reason is that there are several mechanisms of aging. In addition, maybe we accumulate aging factors not only in fluids but also in the extracellular matrix. Because of the sheer volume of the ECM, aging factors hiding there may have an even more pronounced effect than those in our blood. In our paper with Prof. Moskalev, we show the deleterious effect of the ECM on aging. If we do not address this problem, we might not be able to achieve maximal lifespan extension [2].
Yuri Khait (participant)

How did you become interested in biohacking?
I developed a deep interest in this field around 2013. Beforehand, I, like most people, clearly realized that I am going to age and die. I led a regular life, preparing myself for old age, doing some yoga. Both my granddads died from heart attacks at an early age, so I started running. I read a couple of articles about aging and transhumanism and felt a click: this is mine! This is what I need. As if a window burst open. I realized we live during the time when we can defeat aging. I decided that from now on, this was my field. I am a manager in IT, computer services, but my interest in doing business began to fade. Now, I am trying to invest as much of my time as possible in life extension.
Following your decision to start supporting longevity research, how did you choose projects to work on?
Five, six years ago, everything was new to me, and I rushed to participate in every project in this field, from shooting a movie to doing mouse studies. First, I founded Longevity Technologies. It is an educational project that uses social networks for promoting news about rejuvenation biotechnologies, not just about biotech, but also IT, transhumanism, and futurism news. It is mostly volunteer based. We already have a lot of subscribers, but we need much more to bring a real change. If anyone out there is interested in supporting this project or in participating in it, we will be happy to cooperate.
Now, I am practicing a more rational approach, thanks to Alexander Fedintsev. He proposed to gauge any potential intervention according to how much it diminishes the risk of death. From this, you can deduce how much it prolongs life. It is the math of longevity. Alexander has several more criteria for interventions, like whether it has a chance to actually slow aging, which is needed if we want to prolong maximal lifespan. An intervention can be weak – one that extends average lifespan but does not affect maximal lifespan, which is all we have now (good healthcare, nutrition, etc.) Our goal is to find roads to a more drastic extension. We are thinking about some new experiments that can potentially extend lifespan and slow aging. We choose projects according to their promise.
How was the idea of a plasma dilution experiment born?
We follow all the developments in the longevity field. Obviously, we read about early experiments in parabiosis, we read publications by Irina and Michael Conboy. The implementation seemed pretty straightforward, and the results looked interesting. In Russia, many treatments are accessible for money. We phoned a clinic and they told us that apart from adding albumin, it is a standard medical procedure called plasma donation. So, it seemed easy.
Tell us how the experiment progressed. What stages did it include?
First, there was a preparatory stage. Fedintsev prepared a huge panel of blood biomarkers. We did these tests twice – before and after the intervention. It included most of the parameters that can be assessed by a blood test, because we had no idea what effect to look for. Next, we went hunting for places to run the experiment and for people to perform the procedures. Logistics were the most complicated part. We did the first panel in one place, the plasma donation in another place, the albumin injection in yet another place, and so on. With a bit more effort, I think it should be possible to eventually do everything in one place.
Do you think the experiment was safe? Did you have any concerns?
In hindsight, the experiment does not look dangerous. It carries the regular risks of a medical intervention involving blood fractions. It is an invasive procedure. There is some risk of individual reactions. Fortunately, we did not have any. We made sure we used albumin from a good European maker.
Were you worried about being subjected to a potentially powerful intervention?
We still do not know how powerful it is. We need more experiments, but I know that without human trials, we will get nothing. We will be treading water with all these mouse trials. Experiments on mice translate badly to people. This is why we do biohacking with ourselves as the subjects. We just do not have a lot of time.
So, the main motivation is to spur progress, since even such small trials can help formulate protocols for future experiments and spark a discussion in scientific circles, right?
Yes, absolutely, this is the case. Now people are saying: “Wow, so it was possible! Great!” Now we can move forward. I hope it will influence scientific discoveries in the longevity field.
How did it go? How did you feel?
I feel great. Since then, my wife got COVID, but I miraculously did not catch it. I do not know if it is the experiment. I and Nikolay fared well. The day of the procedure, we felt no fatigue, even later in the day when we should have been exhausted. Maybe it was the euphoria.
Do you plan to expand this project? We have a lot of people in our community eager to experiment on themselves.
We do not plan to assemble a large group. We have added this experiment to our interventions portfolio, and we will keep studying it. Let other people decide for themselves if they want to go through it. There are certain risks. But we know that in Russia, there is a study of this procedure being planned with many participants.
Did you discuss the experiment with your family and friends? How did they react? Were they supportive?
It is complicated. My wife is a medical worker. She understands the risks involved in procedures like this. So, she dissed me, and she keeps asking me to stop doing it. I did not share it with many friends, since they would not understand; it would seem strange to them. They think I am a pretty healthy person anyway, so why do it? But there was also support and interest in what we do. Life extension is my personal goal, although I want it not for me personally but for humanity as a whole. I will keep doing it, and there probably will be more experiments we will have to perform on ourselves, because somebody has to do it.
Never quit!
Never quit, exactly. Keep plowing forward. I want to stress that we know what we are doing, and we always try to minimize the risks. We do not want to sacrifice ourselves on the altar of science. Biohacking demands a sober view on what you are doing and a good understanding of it.
I think you have found a good approach – small experiments that serve as proofs of concept and attract investors’ attention.
That is exactly the point. There are many academic studies that are mired in scientific bureaucracy. If only we could find a way to circumvent it – like with COVID, which made many people realize we need to act fast. Suddenly, it was possible to break some rules and produce vaccines faster. We can do the same in the longevity field.
Anything else you would like to add for our readers?
I would like to speak from my business experience. Many people think that the main problem of longevity research is a shortage of funds. I respectfully disagree. The problem is that there is a gap between the people who have this money and the scientific community. People with money do not get enough information about where to invest, where more potential is hidden. There are not a lot of good projects around. When someone invests, there is a good chance that the money will be spent ineffectively. Then, the investor becomes disillusioned and will probably abandon this field. We do not ask for a lot of money. We try to make small steps that do not require huge investments but still hold some practical value. We think this is a good way to get results and to translate them into more large-scale studies. We do not think shortage of money is the problem. There is a lot of money in the world; it is just not allocated properly.
Nikolay Sidorov (participant)

How did you get into biotechnology and life extension?
I had led a pretty regular life by the time I stumbled into biotech in around 1996. Probably, my education played some role – I am a trained therapist, among other things. I studied biology in university. In 1996, I founded a small company that produced bioactive dairy drinks. At some point, I decided that I alone am responsible for my life. My body is my own project. My career and everything else will lose any meaning if my body fails. I realized I need to care for this casing that carries my brain, my self. First, I got into mysticism, then into religion, because they both promise eternal life. Then, I “upshifted” to pseudoscience. Various spoofs like magnetic water. Then, I began forming an opinion about the “four levels” of biohacking. First, you get lured by people who consciously fool you. Then, there is mysticism. Then there are people, scientists who believe in what they do, but what they do is not working, it is false, outdated like the amyloid theory of Alzheimer’s or resveratrol from grapes. Then, I got into biohacking. I think my main biohacking skill that I acquired during those years, by traveling from one conference to another, by educating myself, is to predict what is solid science and what is not with around an 80% success rate, to sift through people who believe in what they are doing but have false beliefs.
When you decided to support life extension, how did you choose your projects?
I have invested 5-6 million dollars into various studies of longevity treatments over the years. I had my own laboratory in Singapore that worked on polysaccharides, then I started investing in virtual online labs. Right now, we do not have a single space or full-time workers, which makes it cheaper and quicker. There are small groups of researchers doing specific tests. I have a guy in Hong Kong. We communicate and work together on the concept. Then, I have a doctor in Rostov-on-Don, I have people in Moscow State University. I might say to them: “Let us see how this or that senolytic works.” First, we receive the compound from China, we make sure this is the compound we need, then we test its safety in a mouse trial in a research institution somewhere near St. Petersburg, then we check certain biomarkers, then we take the compound, and, finally, we check the biomarkers again. My procedures and results do not comply and do not have to comply with FDA criteria. FDA’s rules are devised so that drugs with very low efficacy could get through all the stages and be sold to the consumer. I am looking for very high effectiveness. If it is high, I do not need to go through all the stages and test on large groups of people. If it leads to more than 25% shift in the biomarkers, all other stages are solely about safety (“do no harm”). For instance, our results on senolytics were enough for me to realize that it is not a silver bullet, that it will not extend lifespan, and so we need to move on. I realized it two years ago. It took me just ten thousand dollars to understand that I do not want to further invest in it. It’s just an example. There are around seven fields of study that I am now keeping clear of thanks to the data that I have obtained. Even if I get a negative result, it tells me not to enter this or that field in the future.
Do you always test on yourself or on other people as well?
First, I test on animals, then on myself and on other volunteers, after we know the safety profile and the mechanism of action.
Why did you think this plasma dilution therapy was worth your attention?
First, it was discovered and studied long ago. I did plasmapheresis in 2002 for detox. Then the equipment had gotten better, and the Conboys appeared on the stage. Before that, people thought that there is a youth factor in the blood of the young. Then, they realized that we need to remove the old age factor rather than add a youth factor. We had read all the materials and concluded that we can combine the two techniques – the old one and the new one, plasma dilution and a specific protocol – and that we can do it to ourselves mostly risk-free. Then, there was logistics – how to do it so that we can circumvent the restrictions. The regular scenario followed: the biomarkers panel, the Conboys’ procedure adapted to humans, and the same panel afterwards. We registered large shifts in the biomarkers. It obviously demands more study, but we immediately published it out to the community of biohackers. We published the protocol so people can do what we did and so we can have a larger discussion. The procedure did us some good. For instance, inflammation markers plummeted.
Did you get any unexpected effects?
We, biohackers, are very adept in feeling our bodies. I can predict my body temperature and heart rate precisely. So, after all the three times I did the procedure, I went into a state like if I drank a few cups of coffee in the morning, only the effect from coffee lasts for several hours, and this state lingered for a couple of days each time. I liked it a lot. I felt very brisk and rested.
You are right about the procedure being widely available. Plasmapheresis is being offered now by clinics in Moscow, for instance, to pregnant women in preparation for childbirth.
There are two types of plasmapheresis – membrane and gravitational. The first one does not cut it. It removes the wrong particles, and among the particles it brings back are possible aging factors. The gravitational one removes the aging factors.
It is good to know.
We have a lot of interesting results, but the procedure only works if you replace at least 50% of the plasma. So, if you weigh 70 kilos, you need to do it at least twice, 700 ml of plasma each time, and inject around 70 mg of 20% albumin solution. Here, I just told you the whole thing.
And now for a personal angle: Did you discuss your experiments, particularly this one, with your family and friends? What was their reaction?
I cannot explain to my family and friends why I do this. To understand the whole thing, one has to put an effort into it and learn. This often does not happen. If a person does not really want to understand, I stop explaining, but some people get interested and ask for more info. Then I am all open. Some of them became part of the community, even participated in several experiments. My main criterion is: if you want to learn, to know what is being done to you during the experiment, then I will educate you. I think people today are too eager to outsource the maintenance of their bodies to doctors of all kinds.
We talked a lot about individual health benefits, but I also want to ask, what do you think about the direction in which longevity research is moving and about the potential of these therapies to extend life not just for a small circle of biohackers but for all people?
The way it is happening now is a natural distribution from biohackers to their inner circle, to friends and families, to people who are willing to understand it and to think independently. There are no secrets: if we will invent something that brings radical life extension, there is no reason to keep it to ourselves. States have ways to get those treatments out to the public. The science behind radical longevity is fairly complicated, which makes people wary about adopting it. States are good at suggesting and even imposing things that are hard for the public to understand. Take vaccines – these are state-wide projects. States are good in distributing them, in promoting them. Of course, I can take my hypothetical invention and give it to the state.
What else would you like to tell our readers?
I want to reiterate that people should be actively participating in their own health decision making. They should know what they are doing and what is being done to them and take it into their own hands. It is possible to achieve such a level of knowledge over a year to a year and a half by taking seminars and lessons. Then, you will learn to separate the wheat from the chaff and to maximize benefits from longevity science.
We would like to ask you a small favor. We are a non-profit foundation, and unlike some other organizations, we have no shareholders and no products to sell you. All our news and educational content is free for everyone to read, but it does mean that we rely on the help of people like you. Every contribution, no matter if it’s big or small, supports independent journalism and sustains our future.
Literature
[1] Conboy, I. M., Conboy, M. J., Wagers, A. J., Girma, E. R., Weissman, I. L., & Rando, T. A. (2005). Rejuvenation of aged progenitor cells by exposure to a young systemic environment. Nature, 433(7027), 760-764.
[2] Fedintsev, A., & Moskalev, A. (2020). Stochastic non-enzymatic modification of long-lived macromolecules-a missing hallmark of aging. Ageing Research Reviews, 101097.