Avoiding Osteoporosis with Alpha-Ketoglutarate
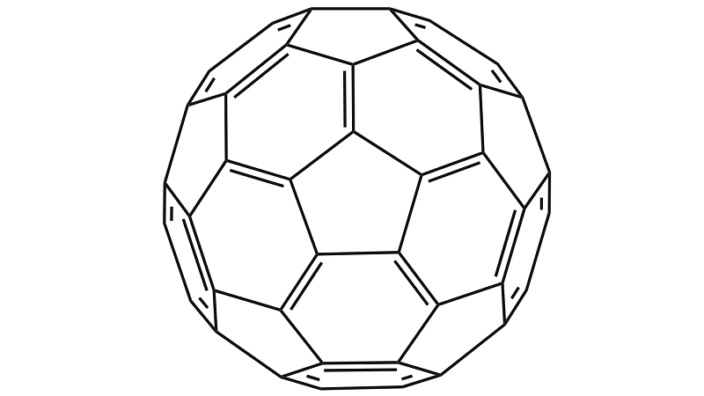
Research on rodents shows that alpha-ketoglutarate could be an effective way to treat or even prevent osteoporosis [1].
Feeling frail
Osteoporosis increases the risk of bone fractures in older people, harming them as individuals and increasing the load on hospitals as the average age of the population rises. Finding ways to prevent bones from weakening with age is thus important for vulnerable people and for the healthcare system in general.
Earlier studies have offered hope that alpha-ketoglutarate (αKG) might prevent bone loss or strengthen weakened bones. Experiments showed that αKG promotes bone development in rats and lambs [2, 3], and it also delays the decline of rapid, coordinated movement in worms [4]. In addition, αKG was shown to have a key role in maintaining the ability of embryonic stem cells to differentiate into any cell type [5]. Based on this, αKG seems like a promising candidate for preventing or repairing osteoporosis, but there haven’t been experiments testing the idea.
Bringing back toughness
A team of researchers in China and the US tested how αKG affects bone mass, bone loss, and healing in mice and rats. They dosed old rodents with two concentrations of αKG in their drinking water and then measured the volume, thickness, and mass of various bones. They found that αKG treatment increased bone mass and density, and they also showed that the treated mice had more osteoblasts and osteoclasts, cells that make and break down bone material. In cell cultures, mesenchymal stem cells, which produce osteoblasts and osteoclasts, from the αKG-treated mice had more osteogenic potential and higher expression of osteogenic genes.
Having shown that αKG can increase bone mass in older rodents, the team then tested whether it could prevent bone loss. They treated adult mice with αKG and found that they lost less bone mass over the next few months than a control group did. Finally, experiments with a “drilling-hole injury” showed that αKG improves bone healing in older rats.
The researchers did some molecular analyses to figure out how αKG boost stem cells to keep bone healthy. They discovered that αKG treatment reduces the levels of certain types of methylation in the stem cells of older mice, leading to increased expression of key bone development genes.
Age-related osteoporosis is characterized by the deterioration in bone volume and strength, partly due to the dysfunction of bone marrow mesenchymal stromal/stem cells (MSCs) during aging. Alpha-ketoglutarate (αKG) is an essential intermediate in the tricarboxylic acid (TCA) cycle. Studies have revealed that αKG extends the lifespan of worms and maintains the pluripotency of embryonic stem cells (ESCs). Here, we show that the administration of αKG increases the bone mass of aged mice, attenuates age-related bone loss, and accelerates bone regeneration of aged rodents. αKG ameliorates the senescence-associated (SA) phenotypes of bone marrow MSCs derived from aged mice, as well as promoting their proliferation, colony formation, migration, and osteogenic potential. Mechanistically, αKG decreases the accumulations of H3K9me3 and H3K27me3, and subsequently upregulates BMP signaling and Nanog expression. Collectively, our findings illuminate the role of αKG in rejuvenating MSCs and ameliorating age-related osteoporosis, with a promising therapeutic potential in age-related diseases.
Conclusion
While this is obviously just a first step, it’s still quite encouraging research. Whether or not αKG ever passes muster and makes it through clinical trials to become an osteoporosis treatment, it will at least serve as a powerful tool to unravel the factors involved. Addressing the burdens of aging will probably involve a variety of treatments to adjust the behavior of a range of different pathways, and αKG seems well positioned to help us tackle issues in bone development and stem cell regulation.
Literature
[1] Wang, Y., Deng, P., Liu, Y., Wu, Y., Chen, Y., Guo, Y., … & Li, Q. (2020). Alpha-ketoglutarate ameliorates age-related osteoporosis via regulating histone methylations. Nature communications, 11(1), 1-14.
[2] Dobrowolski, P., Tomaszewska, E., Bienko, M., Radzki, R. P., & Pierzynowski, S. G. (2013). The effect of dietary administration of 2-oxoglutaric acid on the cartilage and bone of growing rats. British journal of nutrition, 110(4), 651-658.
[3] Harrison, A. P., Tygesen, M. P., Sawa-Wojtanowicz, B., Husted, S., & Tatara, M. R. (2004). α-Ketoglutarate treatment early in postnatal life improves bone density in lambs at slaughter. Bone, 35(1), 204-209.
[4] Chin, R. M., Fu, X., Pai, M. Y., Vergnes, L., Hwang, H., Deng, G., … & Hu, E. (2014). The metabolite α-ketoglutarate extends lifespan by inhibiting ATP synthase and TOR. Nature, 510(7505), 397-401.
[5] Carey, B. W., Finley, L. W., Cross, J. R., Allis, C. D., & Thompson, C. B. (2015). Intracellular α-ketoglutarate maintains the pluripotency of embryonic stem cells. Nature, 518(7539), 413-416.