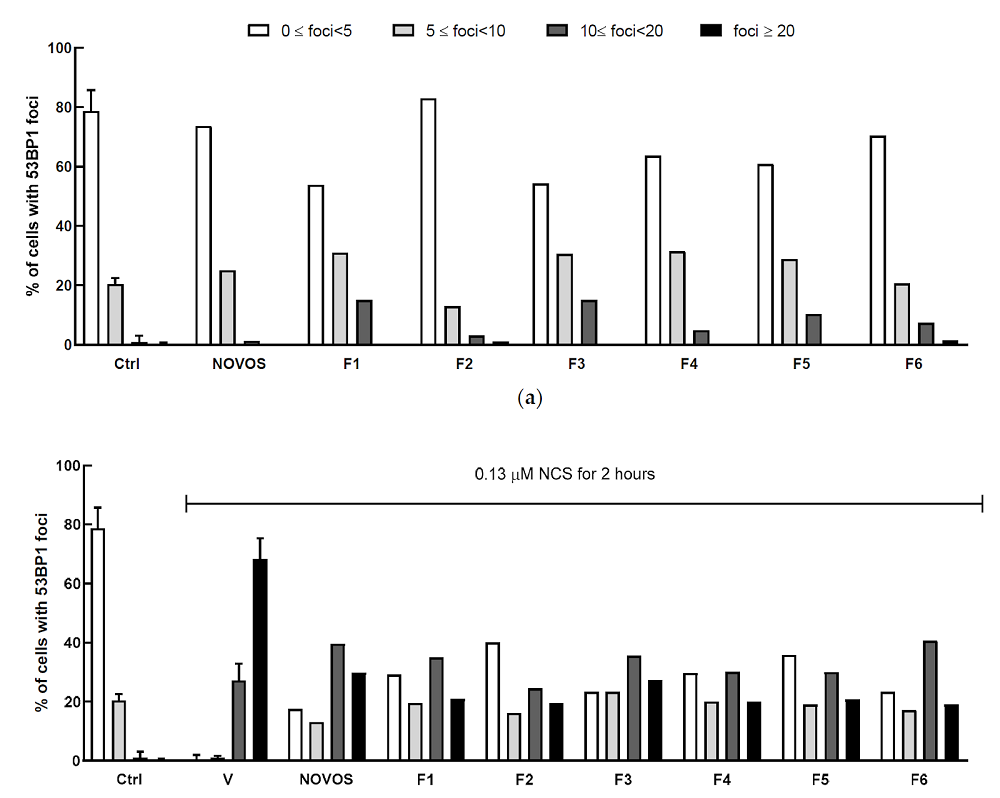
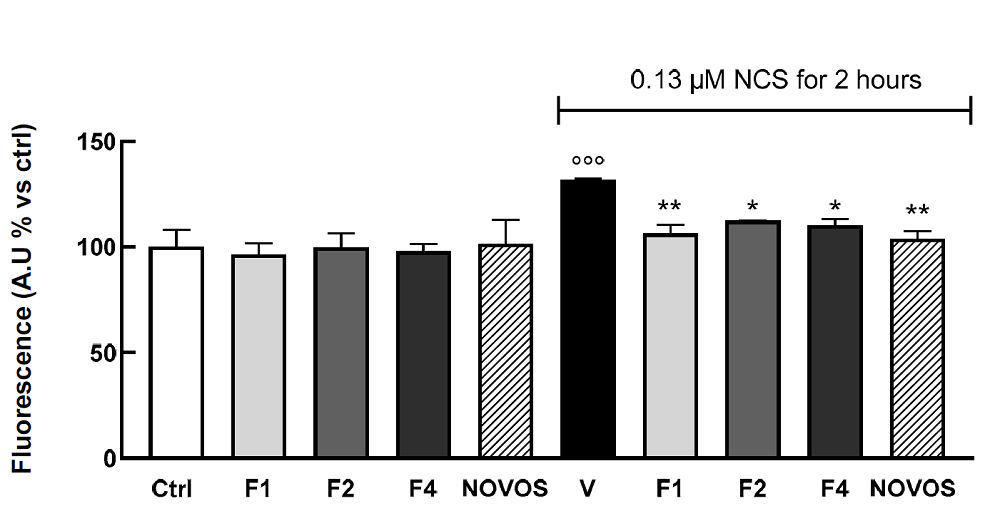
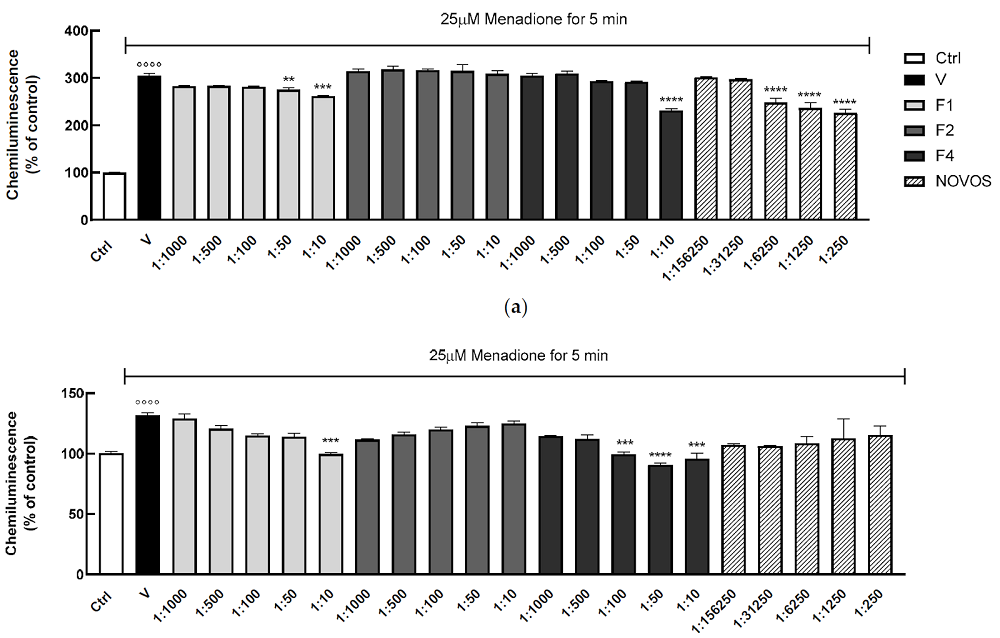
Anxious about being late and distrustful of LA’s afternoon traffic, I got to my destination quite a few minutes early and found myself in a quiet middle-class neighborhood. The house I was looking for did stand out from the rest but rather modestly so.
A week earlier, at a longevity-related event in the Bay Area, I had met Bryan Johnson, the celebrity founder of Blueprint, “the most measured person in the world,” and a self-described rejuvenation athlete. I interviewed him last year, and it was a perfect moment to ask for a follow-up interview.
After we chatted for a while, Bryan said: “Hey, you should come to one of my Don’t Die Dinners.” This is something Bryan has been doing for a couple of years now, with the guest list sometimes featuring first-tier celebrities such as the Kardashians.
Don’t Die is a concept or philosophy that Bryan has been developing. It shares some premises with Ray Kurzweil’s theory of singularity, which states that due to the ever-increasing pace of technological progress, a future is near that is completely unimaginable – and hence, unpredictable from our current standpoint. How do we prepare ourselves for such a future? How do we ensure our survival as a species? How do we find purpose?
Bryan thinks he has at least a partial answer. When we sat down for that follow-up interview in a secluded poolside patio, some half an hour before guests were supposed to start arriving for dinner, he tried to explain it all to me.
You think big. Your philosophy deals with monumental things like preparing humanity for the age of AI, but your answer is, basically, “exercise, eat healthy, sleep well.” Isn’t it a bit anticlimactic? How does all this work together?
If you take a few moments in history, like the day the atomic bomb was demonstrated, or the day before the Wright brothers’ flight, or the day before ChatGPT launched, and ask people what tomorrow would look like, they will describe certain models of the world. And then tomorrow happens, and everything’s different. Everything you thought to be the case has now changed, and that’s my fundamental premise.
Basically, it means that we’re on the eve of everything changing beyond recognition, even if today looks normal and tomorrow feels like it’s going to be normal. We think this is going to be the case, but it’s not. Sometimes, things just change dramatically.
So, in this event of a big change, what’s in our power is not to know what’s coming because we can’t predict that, it’s to get the philosophy of existence correct. Don’t Die is the philosophy, and the way it manifests itself is in our individual actions.
When I started this project, I thought, okay, we’re currently facing several existential crises. We have climate risk, AI risk, we’re at each other’s throats with nukes and biowarfare. We’re in a dangerous spot as a species. Many things could either collapse society or eliminate us.
How can we address any of these problems if a vast majority of people do not feel empowered to do anything? What can they do about AI? What can they do about climate change – recycle Amazon boxes?
Don’t Die is a way to say you’re actually not powerless because the entirety of our existence is the collection of all of our behaviors. And as irrelevant as exercise, sleep, or diet can feel, they’re not. By your decision not to go to bed on time, not to exercise, you’re basically stamping with approval everything that’s going on today. You’re participating in this “die culture.”
So, Don’t Die is meant to say that we have power, we can do something. These small actions we take cause this cascade of follow-on effects.
Do you want people to act differently or to think differently?
Both.
So, what you’re saying is that acting differently makes you think differently?
Exactly. If I step into the Don’t Die world, this means that I’m going to start eating well, exercise, become aware of whether my foods are clean or not, of what happens to my body.
And then, this might make me become aware of what my child is eating at school. Why are you feeding my child pizza, canned vegetables, and chocolate milk? How does that make any sense at all?
It’s this cascade: once you internalize it for your own life, you start seeing the world through this new frame. I believe that doing the basics of health and wellness can lead to remapping people’s understanding of the world. They start seeing everything through this new lens.
If you have some sort of respect and sympathy for your own body and health, you might project this mindset onto the outside world, including nature, right?
Absolutely. We treat planet Earth the same way we treat our body. We go YOLO with our bodies – and then we do the same to Earth, we pollute it with abandon, and so on. So, yes, when you begin caring for your body, you then, by proxy, start treating everything differently – your child, his school, politics, all of it.
Do you speak from experience here, or is this purely theoretical?
Definitely, it’s my personal experience too. I now see the world entirely through this lens. I can’t look at anything and not see this construct.
“Don’t Die” sounds great, but we don’t know if longevity escape velocity is even achievable, not to mention how far away it is. So, you talk about not dying, but then you don’t really give people a blueprint – pun intended – for not dying. Because Blueprint doesn’t seem like a recipe for not dying at this point, just a recipe for being biologically younger.
I hope you’ll find answers in our tonight’s Don’t Die conversation. A person only acquires an understanding of Don’t Die after a two-hour-long conversation. In fact, I have never succeeded in getting someone to understand Don’t Die without this talk.
How, then, do you plan to scale your philosophy? How will it take over the world?
I wrote Don’t Die host guidelines. If you want to host a Don’t Die Dinner, there’s a 35-page document and it plays out the entire event. I want eventually a million such dinners held by people all over the world. You’ll see it tonight, I don’t want to spoil it for you.
Sure, in this case, let’s move on to some practical things you did with Blueprint recently. For instance, I think it’s interesting that you cut your caloric restriction by half, from 20% to 10%.
Yes, the hypothesis was that caloric restriction is positive, but how much CR is optimal? We started with 20%. We had 1,950 calories, and my RDA (recommended daily amount) is 2,500. As a result, I got really skinny. And we wondered, do I really need to be this skinny? Is 10% CR or no CR at all just as efficacious? So, we went from 20% to 10%, and we lost nothing on the biomarkers.
And what about fat content? I think it was about 6%, and you thought at that time this was optimal.
I did put on about 18 pounds, gained a lot of muscle, but also a lot of fat. And my biomarkers are still the same. It’s an interesting experiment, and the question is, what if I went all the way up to 2,500 calories, zero CR?
Your staying just as healthy despite gaining some fat actually aligns well with a lot of previous research. And then, there were those blood transfusions everyone’s talking about.
Yes, I did six of them. I had a donor who was eating a Blueprint diet. I did five total infusions from that donor and then one from my son. And over that time course, we couldn’t detect any changes in my biomarkers. That’s not to say there weren’t any positive or negative effects, but we just couldn’t find them.
Or isolate them?
Correct. However, at that time, we weren’t doing many new things. I did this once every three weeks, so it was a pretty short window of time. But when my father did just one infusion, when I gave him one liter of my plasma, and he removed 700 milliliters of his plasma, his speed of aging measured by DunedinPACE clock went down from the age equivalent of 71 to 46 immediately. A dramatic effect. And this is about the same effect size you see in rats; it mapped pretty closely.
We know that older and less healthy animals benefit the most from many anti-aging therapies.
Exactly.
Continuing with therapies, a few months ago, you went to Vitalia to get a follistatin gene therapy. How did that go? Do you have any preliminary results?
I’d say, three things. One, my follistatin levels increased by 160% fourteen days after the treatment. That was their hypothesis about C-max (maximum concentration). I don’t know if that’s true, because we only did one measurement. So, at least a 160% increase.
Then, my muscle mass. It was already in the top one percentile for optimal, and I got an additional increase of 7%. And finally, my speed of aging dropped from 0.7 to 0.64.
And is this because of follistatin?
That was the only thing that changed.
Blueprint going commercial surprised a lot of people. How did that happen?
I had no commercial intent when I started Blueprint. It was simply an exploration – what can a person do in this field? I didn’t expect it to go viral, but it did, and the number one thing people said is, make it easy. “I see your page, you have all these supplements, drugs, treatments, it’s so complicated, I don’t want to do it, it’s too much.”
Sort of, “Simplify this and take my money”?
Yes. “Just make it easy.” And I got this request from everybody. I didn’t want to be in the food business. It sucks. Low margins, a lot of headaches, everyone’s doing it. And I sat on it for six months. After Blueprint went viral, I didn’t do anything for six to eight months. I just tried to evaluate my next steps because once you start something, you own it, right?
When we were doing Blueprint, we would test all the supplements, all the food I was eating, and we found that most of the food was alarmingly dirty. I’m not sure you’re aware of how dirty food is. Even in supplements, you don’t always get what’s on the label.
We tried to tackle this problem ourselves to see if we could buy ingredients ourselves, do testing, and build the whole supply chain. And my friends were like, “Can I have it too?”
This is how you source your supplements, too?
Yes, I started sourcing everything myself – the olive oil, the chocolate, all the supplements. For instance, we would buy raw batches of lentils and test them for everything. We searched the world over for clean lentils and couldn’t find any, even the ones that were labeled organic. Everything was alarmingly dirty.
Finally, I said, okay, people want it to be easy, this is legitimately a major problem. What if we could just be the source of every calorie a person wants to consume? But I’m definitely not doing it for money, otherwise I wouldn’t be doing it. There are so many things that make much more money.
Have you then thought about publishing more data or more detailed data about all your supplements and food and how they are cleaner?
Yes, I actually posted a video today on X. We’re doing it. I basically went to the grocery store, I took ten dark chocolates, and measured them for heavy metals and flavanols. So, yes, we’re starting to share this publicly.
When we talked last year, you said you didn’t want to invest in or sponsor fundamental longevity research because you have your own way of contributing, of doing things. Is this something that has changed?
No. I learned this when I was investing in deep tech. The hit rate in deep tech is very low. It’s very hard to make money in deep tech.
So, you don’t want to bet on discovery. You want to bet on companies that build picks and shovels for the entire industry. That’s why I bet on Gingko, for instance. They are building the ability to read and write DNA at a lower cost.
Basically, we think that synthetic biology is here, so, everyone will want to do all these things with biology, and the critical element we need is low-cost read and write capabilities.
If you’re trying to go for a specific therapy or indication, you only have one shot on target. You have your molecule, your FDA trials, and you either win or lose.
So, you prefer to invest in universally needed tools?
I’ve been trying to think methodically: with the limited capacity I have as a human, how do I find the biggest fulcrum? I could certainly fund non-profits and companies, but instead, I’m saying, why don’t I try to build the basics of sleep, diet, and exercise for hundreds of millions of people and a philosophy for billions of people? To me, that’s a bigger fulcrum than a single drug, or a single non-profit, or a single study, and so on.
I’m inherently an entrepreneur trying to find arbitrage in systems, I like arbitrage. And I think that the world’s systems are breaking, and new systems will have to be better. I think that it’s better to make money out of the gate than to bet on these ten-year-long things, and it’s better to empower millions of people to be part of the ecosystem than a few.
I know that people criticize me: “You’ve got money! Do this, do that!” They’re trying to be capital allocators. What they don’t understand is that they’re not me, I’m not them, I don’t think like them, and they don’t even understand my vision, what I’m trying to do.
Have you been talking to other high-net-worth individuals about longevity? I’ve heard from many people that multimillionaires and billionaires are notoriously reluctant to join our cause.
Yes, I have talked to many, and I think they get it but in a particular way. They don’t think we’re going to stop aging imminently, but they agree with the primary thesis of my argument.
I think what you’re saying is that they’re discouraged, there’s no movement. They don’t change their habits, they don’t invest in biotech or aging, and it seems like it makes no sense. Your only real asset in life is your existence. Why would you just die without a fight? You can’t take your money with you.
Basically, they’re playing the “immortality of accomplishment” game, chasing status and power. What I’m saying to them is, hey, I understand that you have your worldview, but here’s a different perspective that’s going to change everything you care about. The world is about to change fundamentally, and that affects everything you’re doing. All of it may be of no consequence.
Now, that has the potential of stopping them in their tracks, giving them this “I can’t just carry on as is now” feeling. That doesn’t mean they’re going to just drop a billion dollars on funding anti-aging research, but they get the idea that everything they care about – all their status, their power, their reputation, is potentially now at risk because of this major change that’s coming.
Is this working?
I’ve had several billionaires who, after we engaged, just said: “I’m in, what do you need?” I haven’t cashed those checks yet. I’m really trying to build a solid community, not to act hastily. I’m trying to build the infrastructure. That’s what I’ve spent the past six months on – how to transform Don’t Die into an actionable plan politically, economically, morally, ethically, socially.
The pudding is served
As we were talking, guests began to arrive, and the growing murmur of voices signaled that we should wrap things up and head back inside. I didn’t mind – after all I’d heard about Don’t Die, I couldn’t wait to see Bryan in action.
He told me in advance that this time, the crowd was going to be unusually large, around 30 people (no Kardashians, though). And what a motley LA crowd it was! Entrepreneurs and physicians, artists and scientists, investors and inventors, and, well, one science journalist. Most were dressed casually, while two or three looked like they came straight from Burning Man (in fact, they ended up inviting everyone to Burning Man).

As guests mingled in a crowded foyer, a modest dinner was served. First, we toasted with shots of Bryan’s olive oil, which he proudly markets (with tongue in cheek) as “Snake Oil”. By then, I had already bought a couple of bottles and formed my opinion: great quality, a bit tangy for my taste.
Then, each guest picked up an earthenware bowl of Nutty Pudding, a staple of Bryan’s kitchen made from macadamia milk and nuts, walnuts, chia seeds, flaxseed, cocoa, berries, cherries, cinnamon, and pomegranate juice. This super-duper-healthy slush has a distinct dusty pink color and an equally distinct taste: yummy but takes a bit of getting used to.
Clutching our bowls, we all stepped into the living room and sat down. Bryan started by asking people to introduce someone else in the audience. This proved to be a great bonding experience, albeit time-consuming. Since people were probably invited by word of mouth, many of them knew someone, so that wasn’t a problem for them. This left a handful of loners, such as myself, but I quickly struck a deal with the person sitting next to me, who turned out to be a nuclear scientist, and we introduced each other.
Do we know anything at all?
Then the action began. Bryan told us that he was going to ask us a series of questions. The first one was whether we’d agree to replace our own decisions with an algorithm if we knew for sure that it guarantees us the best physical, mental, and spiritual health we can imagine. This was a clear nod to Blueprint, which is touted by Bryan as the algorithm that should supplant your faulty decision-making mechanisms when it comes to eating, sleeping, exercising, and other types of body maintenance.
This got the room divided, with people either objecting to the idea that an algorithm can know more about our well-being than we do or pointing out that it was a trick question: if the algorithm is going to make us happy by definition, there was no reason not to succumb to it. After all, it cannot make us simultaneously happy and unhappy.
Next, Bryan invited us to think about how people from the 25th century would view our morals, ethics, and social norms. The audience generally agreed that our descendants would find objectionable our penchant for killing our bodies, our brethren, and other animals while being hopelessly divided by politics and religion. However, it was also quite clear that we had zero idea about who those descendants may be and whether they will exist at all.
Then, Bryan told us about his son, a college student. For the entirety of human history, he said, things kept changing slowly enough for people to be able to imagine their life course. Just a couple of decades ago, the path, the possibilities were understandable – you could, at least in theory, choose an occupation, learn the trade, earn your living, and retire in due course.
Today, with the advance of AI, things have changed fundamentally. What should his son study, Bryan asked? How should he chart his path?
Predictably, no one in the audience could conjure a solid answer. Bryan used this and previous questions as a segue to talk about our greatly increased inability to predict the future. Many, if not all, of our dearly held concepts might not survive in a post-AI world.
For the entire evening, Bryan subtly led us to this conclusion, showing that people tend to “consult their knowns,” but this method isn’t effective enough in an ever less-predictable world. Even thinking from the first principles won’t cut it. Instead, we have to think from the “zeroth principles,” that is, assume that we know next to nothing, and imagine something entirely new.
Don’t Die, and find out
What should we do, then? According to Bryan, Don’t Die is the solution. To be honest, I still can’t entirely wrap my head around it. Does it mean it’s wrong? Not necessarily. A hard-to-comprehend idea can be a genius one.
However, I feel that Don’t Die philosophy is still a work in progress (something Bryan himself confirmed to me), a star-forming cloud of ideas, some of which seem intriguing and profound, while others are sort of headscratchers for me at this point.
One idea that aligns well with the longevity field is that of the sanctity of life and the immense value in extending it. Bryan’s twist on it is that cherishing life, consciousness, and the body that contains it provides a comprehensible goal most people can unite behind.
“This is our sole known priority,” Brian said. “The only thing we all agree upon. It’s not a solution to everything, but it’s a framework, like democracy. Ideologies cannot provide the same level of unity. It’s our only common identity when we can’t plan forward.” His book Don’t Die: Dialogues adds, “the ‘fountain of youth’ thing is a clear and good goal because, at the very least, we get that goal. We understand it. We can define it.”
Bryan sees the world’s reaction to the COVID pandemic as a manifestation of this common goal that we all share deep inside, even if, ostensibly, it might not always align with our ideologies or religious beliefs. Again, this weird dualism, where people value life and good health but do not translate this into an anti-aging sentiment, is well known, and Bryan is trying to bridge this gap.
Bryan thinks that learning to trust algorithms, like he does with Blueprint, can potentially free up so much time and energy “to explore new frontiers” that the impact will be comparable to the invention of fire, which uniquely sped up human civilization. We would both be more mentally fit and have more time on our hands (indefinitely more if we can figure out how to stop and reverse aging).
As to abandoning our beliefs and embracing the unknown, I actually agree that not only does this nurture open-mindedness and curiosity, but also, somewhat surprisingly, quells that nagging dread of the future. We’ll just have to wait and see, right? We’re an inquisitive, fearless, and inventive species that has extricated itself from all kinds of tight places before. But to be able to stick around, we’ll have to stay alive.
On the way, we’ll also have to learn to trust algorithms that will be taking away more and more of our tasks to the point of threatening our very relevance. Building machine minds is a dangerous game, but humanity is bound to play it. There’s just no other way – again, something that most aging scientists would agree with (aging is probably just too complex for the unassisted human mind to unravel).
As I approached Bryan to thank him for the evening, it was way past his bedtime. “See,” he said, smiling, “I do sacrifice things sometimes when I feel it’s worth it.”
He asked me what I thought of the dinner. “It was very thought-provoking,” I said frankly as I shook his hand. “For me, this is one of the best feelings in the world.”
Did the dinner turn me into a Don’t Die warrior? Probably not, but on the other hand, I’m already an ally. Is Don’t Die the best way to promote “the longevity cause”? I don’t know. But it’s not a bad way, either. It’s just Bryan’s way.
We would like to ask you a small favor. We are a non-profit foundation, and unlike some other organizations, we have no shareholders and no products to sell you. All our news and educational content is free for everyone to read, but it does mean that we rely on the help of people like you. Every contribution, no matter if it’s big or small, supports independent journalism and sustains our future.