To many people, Peter Diamandis needs no introduction, and to the rest, it’s going to be a long one. To put it as concisely as possible, Peter is a serial entrepreneur and investor in several fields, from commercial space flight to longevity biotech; a visionary speaker and author of several best-selling books, including Abundance, The Future is Faster Than You Think, and Life Force; creator of the world-famous XPRIZEs, including the recently announced massive XPRIZE Healthspan; founder of the Singularity University… I could go on and on, but I’d rather let Peter do most of the talking.
You have a very interesting biography, and your “first love” was space travel. How did you get involved in longevity?
Yes, I grew up passionate about space flight, on the heels of Apollo and Star Trek. But my dad was a physician, and my parents expected me to go into medicine as well. At some point, I said, okay, I’ll do it, because it’s a way of becoming an astronaut.
Long story short, I went to medical school, and by the end of it I was already running a few companies, mostly in the space business. I remember watching a TV show on long-lived sea life – bowhead whales, Greenland sharks. Then I had this thought: if they can live that long, why can’t we?
I was an engineer and a medical student, and I said to myself that it was either a hardware or a software problem. Anyway, we’re going to be able to solve it in the future. That was the first time I really started thinking about that.
The second time I started thinking about longevity was when I realized that the commercialization of space flight was moving a lot slower than I wanted it to be. And I thought that if I wanted to make my dream of going to the stars come true, I would have to live a lot longer for the technology to get there.
Sounds like, initially, longevity was a means to an end for you, but today, your involvement in this field is deep and sprawling, correct?
I consider myself mostly an entrepreneur. At this point, I’ve started 26-27 companies. About 12 years ago, I reengaged with longevity by starting Human Longevity with Craig Venter and Dr. Bob Hariri. About at the same time, I started to realize that technologies required to reengineer the human body, its hardware and software, were coming online.
I’d become enamored with the notion that exponential technologies are going to give us the tools to fundamentally understand why we age, and how to slow, stop, perhaps even reverse aging. And there’s nothing of greater value.
I think if you’re healthy and love life, you will want to see as much of it as you can. People who are in pain or have disabilities or chronic disease towards the end of their lives don’t have a desire to live an extra 50 years. But if we can rid them of that pain, lack of energy, chronic diseases, cognitive decline, who would not want to continue that vibrant life?
So, I became more and more interested in the longevity field, and I started building companies in it, and as I did in the space industry 30 years ago, I approached this from multiple directions. I started a venture fund, BOLD Capital, and we’ve steered it towards longevity. BOLD Capital III, our third fund is, probably, two thirds biotech investments. We’ll be announcing shortly the first close of Bold Longevity Growth, which is a growth venture fund in partnership with Sergei Young.
The second thing I did was the educational push. I run something called Platinum Longevity Trips. I’m scanning about 500 companies a year through my venture fund, through XPRIZE, through other means. We choose the top 50 companies, and we run an adventure trip to go and visit those, meet with the CEOs. I bring about 80 family offices, venture capitalists and CEOs. It’s a five-day deep dive. Basically, I learn as much as I can, and I teach as much as I can. There’s also my Abundance Summit every year, and longevity is a very important part of that.
The third part is the books I’ve written on the subject. The fourth is the 101-million-dollar XPRIZE Healthspan. I’m a contributor towards it. Money came from Chip Wilson, the founder of Lululemon, from Hevolution, and from several people from my abundance community.
Finally, the last part is starting companies, such as Fountain Life, Celularity, and Vaxxinity.
We’ll talk about it all, but first, it’s interesting that you moved out of the space field because it was moving too slowly. Do you ever get the feeling that the longevity field is moving even more slowly? How optimistic are you about it?
The longevity field has a lot of parallels to commercial space flight. In the early days, a lot of it was based on faith. There was this huge dream, big potential, but it was in the realm of science fiction. And there were all those crazy entrepreneurs trying to do things.
In the space business, it was either government funding or this unique class of commercial – comm satellites and military, but it wasn’t the entrepreneurial approach.
In the longevity field, it’s been similar – government-funded research and the pharma industry focused on chronic diseases. But conversations specifically about longevity and slowing or reversing aging were always viewed as fanciful and crazy.
But now we’re seeing this transition, just like it was with space, where the entrepreneurs are coming in. These are small, agile teams, backed by venture capital and these exponential technologies, and they are beginning to really accelerate the field.
This makes me extremely hopeful. For me, the two biggest businesses in the world, the two biggest opportunities are AI and longevity.
One of your principles is “if you want to become a billionaire, you should help a billion people”. With longevity, we’re talking about eight billion people.
Yes, and I think, what an amazing beneficial coincidence this is – all eight billion of us have the same disease of aging, so anything that helps a wealthy billionaire will help the poorest individual as well.
People ask me all the time whether we’re talking about therapeutics for just the wealthiest, the top 1%. But I don’t think that will be the case.
Maybe for the first few years. But just like mRNA vaccines, which essentially are gene therapies, became mass-produced and dropped to a cost of a couple of dollars per dose, it’s likely that we’ll see the same happening with longevity therapeutics – they will be mass-produced, and the price will drop precipitously.
But to be honest, your current flagship longevity company, Fountain Life, just like Human Longevity before, and your ultra-exclusive trips – all of them sort of reinforce this stereotype of longevity treatments for the rich only. How important is democratizing longevity to you?
Very important. And those businesses have to be viable on their own to succeed. So, yes, the longevity trips are expensive, they’re 70 thousand dollars a head, but the point of those trips is to connect these high-net-worth investors to the longevity space.
I take the data from those trips, and I put it out for free through my Moonshots podcast, through my books, and my blogs. I can’t afford having ten thousand people visiting these labs, but the information I get from them isn’t locked up forever, it’s disseminated.
With Fountain Life, we’re getting ready to launch a lower-tier product, and the technology is demonetizing. Believe me, we want to run that demonetization curve. The cost of imaging technologies, AI as your diagnostician, all these things are rapidly demonetizing.
As with every technology, like the cellphone, at first, it’s expensive and paid for by millionaires and billionaires. Eventually, it starts working really well and being available to all. So, now, we have a 40-dollar smartphone with massive amounts of free content on it, and it works a thousand times better than the first cell phones did thirty years ago.
That will also be true in all these areas, especially in diagnostics. It’s just not there yet, but it will be, probably in the next five years.
I understand the idea of drawing private capital in with those expensive, bespoke, concierge-style solutions, which brings me to another question. You’re a sort of a “billionaire whisperer”.
Billionaires sit on vast resources, but they don’t seem to be ready to massively invest in longevity yet, even though that would be a very natural thing for them to do, many of them not being that young. What’s your experience discussing longevity with them?
I’ve had very blunt conversations with many of them. When I was working to raise the 141 million dollars for our healthspan prize, we eventually got two billionaires to commit to the prize.
But the situation I want to tell you about is when we set up several virtual meetings, where I sometimes had a dozen billionaires on Zoom with me, most of them in their fifties to eighties. In one year, I met with probably 30 of 40 billionaires in that fashion. In total, I pitched well over 50 or 60 during the course of the competition.
My message to them was: “Here’s the deal – you don’t pay the money until it’s won. You do pay a small amount that runs the prize, but the majority is pledged. And when it gets won, you will have early access to these benefits.”
I would have imagined that all of them would have been fighting to be in a position to donate. I donated a significant amount of my wealth. It’s my largest philanthropic donation ever. And it was crickets. The vast majority of them said “no”.
This made me realize that many of these individuals got there by looking at money as a mechanism to make more money. And they don’t give the longevity business the credibility it deserves as an investment opportunity yet.
Today, people are pouring money into SpaceX, which was a crazy idea 15 years ago. So, it’s a challenge. At some point, maybe in two years, maybe in five, but no longer than ten years, the bit will flip, and people will be clamoring to invest. But it’s not there yet.
But it shouldn’t be just about the money. This is literally about life and death.
You’re absolutely right, but that’s just not the way they’re viewing it. You could say that all of us put our money in three different buckets.
There’s a bucket for money that will be used to make more money. Another one is for money that will be used for family, enjoyment, vacations, and so on.
And the last one is for helping people. Unfortunately, for most people, that last bucket is the smallest by orders of magnitude.
I think it’s not just about philanthropy. It should also be driven by egoistic considerations. Maybe there’s a place for a fourth bucket, where you help humanity, but you also help yourself. After all, today, death is pretty inescapable whether you’re a billionaire or a construction worker. Even more so than, say, climate change.
It seems obvious to me. Why aren’t people acting in their own self-interest? This thing should hit every bucket. I’m going to invest in longevity because it’s going to help me, my family, and humanity, and it will also allow me to feel better, to have a happier life.
And it’s also a huge business opportunity, right?
The only reason that’s not hitting is that people don’t believe it yet. We have been so indoctrinated about the inevitability of death!
I try to change it, by writing books about this. My mission is to help the public create a longevity mindset, which is based on the realization that the technology to enable us to extend the healthy human lifespan is coming online now, in the decade ahead.
And if you believe that in the next ten years, we’re going to significantly extend the human healthspan, and you can have access to it if you’re in good health, then, logically, you’d want to do everything you can to remain in the best health possible.
I want people to change the way they think about healthspan and longevity and what’s possible for them. Because your mindset is the most powerful tool that you have. If you believe it, you’re going to change your behavior.
As a serial investor in various fields, how do you see longevity biotech today? What is your investment philosophy, favorite subfields?
I have an investment team that does due diligence on companies. I make some seed investments, and I’ve taken advisory positions in a few companies.
I do feel that cellular medicine and epigenetic reprogramming are critical. I’m working in “a bridge to a bridge to a bridge” paradigm, something that Ray Kurzweil talks about, which is making sure I’m tackling the near-term things that will buy me the next 10, 20, 30 years, which then buys me the next 50 years. This is why immune health, cognitive function, and musculoskeletal function are things I’m always focused on.
Also, companies that create a revolutionary change are going to be powered by AI systems in one shape or another. This is why I’m very excited about what AlphaFold3 has done, and where DeepMind CEO, Demis Hassabis and his team want to take it in the future, which is AI modeling of an entire human cell, and then AI modeling of you and me.
I’m also always looking at the entrepreneur, this is crucial: am I excited about them? How passionate and dedicated are they? How are they leveraging exponential tech? What is their primary near-term focus?
Importantly, most companies have the potential to go after 30 or 40 things, but it’s narrow focus that determines the winner early on. So, I make sure that the entrepreneur knows what the first thing they’re going to do is, what’s going to drive revenue.
We’ve had this incredibly decimating biotech winter over the last two-three years, which has been crushing, and that’s because so many companies went public before they had consistent revenues or any profits. I think revenue is king. How do you build something that has near-term revenue?
But quick revenue is sort of antithetical to rejuvenation biotech.
And yet, there are companies like Rejuvenate Bio which uses gene therapies in dogs. This is a marketplace that’s easier to get approval for, and there’s a large population of buyers. I’m fascinated by business models like that. There are several pet-based business models, and most of them translate to humans.
Companies where you have to constantly raise money for five or ten years only work sometimes, if the financial markets are moving in the right direction.
What do you think of potentially high-impact areas such as organ and tissue replacement that might allow us to “hack” aging without really understanding it?
Again, this is all about living long enough to live forever. To go back to Ray’s theory, he talks about the three bridges.
Bridge 1 is the current technology.
Bridge 2 is technology that’s coming online, and that includes organogenesis and replacement – things that are going to keep you healthy and going.
Bridge 3, which in his mind, is nanotechnology.
If I had the ability to grow a backup set of organs and have them in the freezer in the garage, why not? It’s an alternative to today’s organ donor marketplace which is insane and cruel to the recipient, given how long the waiting lists are, so it’s obviously important.
Tell me about XPRIZE Healthspan, and how it became the biggest XPRIZE of all time.
XPRIZEs are meant to be competitions that ignite people’s imagination, that get them wondering, “How would I do that?” It’s about giving them a stretched goal with a clearly defined finishing line.
Evolutionarily, we’re designed to compete. We do our best work when we compete for a mate, in the Olympics, in business. Why not use that drive of competition to get people to work on solving the world’s biggest problems? But, XPRIZEs also do something else: they credential a field. They sort of say: “This is real, worth doing, achievable.”
Aubrey de Grey approached me about a longevity prize just after the original XPRIZE for space flight was won. But we could never define the goal, because a prize for extending human healthspan or lifespan by 20 or 30 years would take 30 years to pay out. It just didn’t seem viable.
A few years later, Sergey Young put up half a million dollars to study the idea. Aubrey was involved again. At first, I said, no, I can’t, as much as I want this, I don’t know how we would do it.
Then there was a conversation with George Church and a subsequent conversation with David Sinclair, both of whom were advisors for this prize from early on. And they said, let’s not talk about longevity, or even about age reversal, but about functional reversal. Let’s look at the restoration of age-related loss of function.
The human body was never engineered to live past age 30. We peak in our late 20s. 200 thousand years ago, you were pregnant at 12 or 13, you were a grandparent at the age of 27-28, and soon, you were no longer of any use to the species. The best thing you could do was die. There was no positive selective pressure for living long, so, we didn’t.
This is why past age 30, we have this slow decline, immunosenescence, stem cell population exhaustion, sarcopenia, cognitive decline – all these things decline slowly till death at a measurable rate.
So, the idea is, basically, screw Darwin, we want to take up the mantle of reversing this degradation. If you’re a team that can show in a population of 65 to 85 that you can roll cognitive function, immune function, muscular function back by at least 10 years, you can win this.
It’s all about getting as many smart people into this mission as possible and about getting the regulators to view aging as a reversible chronic disease. One of the things we did with the original space flight XPRIZE is that we got the regulators to create a structure for private space flight carrying humans, which did not exist before. These competitions have the ability to do that.
It’s a global competition, and if it’s a success, we’re going to hit the headlines, attract many more funders, get regulatory support, and change public opinion. We will maybe prove not that aging is not inevitable and that we can fight back against the dying of the light. That we can extend our healthspan. And that’s one of the most important things we can do.
Let’s talk about your two books on longevity – Life Force and Longevity: Your Practical Playbook. How are they different, and what did you try to achieve with each one?
Life Force, which I wrote with Tony Robbins, was a New York Times bestseller. I’m very proud of this book. But it’s a long book, 700 pages! And no matter how good it is, many people won’t read 700-page books.
I wanted to author something that would be consumable, and this is how the Practical Playbook was born.
It started originally because people would ask me, what do I do for my own health – what do I eat, how do I exercise, and so on. It started as a ten-page Word document, and then it grew and grew, and now, it’s a 120-page book.
People can get a digital digest of the book for free, because my mission is to get those basic ideas out into the world.
I’m updating this book every year, and the 2025 edition is coming out in December. So, it’s a living book, so to speak – everything I’m learning, I’m trying to make available and easily accessible.
You already mentioned Fountain Life and Celularity, two of the companies you personally founded that are longevity-related. Tell me more about them.
Fountain Life is a company I started with Tony Robbins. I serve as executive chairman. It’s a company focused on providing the most advanced diagnostics in an integrated fashion, analyzed by a functional medicine AI and a team of AI physicians and care specialists.
It’s a membership model. The goal is for people to come for a full-day “upload.” It takes about six hours where we generate 150 gigabytes of data about the patient. This includes things like full-body MRI, brain vasculature MRI, coronary CT, lung CT, genomics, DEXA scan, metabolomics, blood biomarkers.
All this is to answer two questions. One, is there anything going on inside you that you need to know about? And if so, should we take care of it right away? Second, what’s likely to happen to you, and how do we prevent it?
I call my annual upload “not dying from something stupid.” In other words, there’s going to be that moment in time when something goes awry, and the very best thing you could do is to catch it at the earliest stage. And that’s the primary function of Fountain Life. I’m very proud of it.
Celularity is a company where I serve as a co-founder and vice chairman. Dr. Bob Hariri is the founder, CEO, and chairman. He’s a brilliant regenerative medicine doctor who ran the cellular medicine division of Celgene, which grew to a 120-billion-dollar juggernaut in the pharma industry. I supported him in spinning out the cellular medicine division from Celgene, which became Celularity.
Celularity supports allowing pregnant mothers and families to bank their placentas, and newborn placentas are basically mined for placental materials. We have placenta-derived T cells, natural killer cells, stem cells, exosomes. The company is focused on regenerative medicine.
You seem to believe that longevity escape velocity (LEV) can be achieved during our lifetime. How do you think this will happen, and in what timeframe?
I do believe that we will reach LEV. This decade will see the convergence of a variety of exponential technologies, in particular AI that will enable us to understand the details of cellular and organismal aging.
Models like AlphaFold already do an incredible job in predicting molecule interactions. What comes after that is that AI will help us to model an entire human cell, and then entire tissues and organs. And then we will begin to understand how to slow, stop, and reverse aging.
We’re having billions of dollars flowing into this field. Epigenetic reprogramming will play a critical role. There will be multiple other steps, from organ replacement to cellular medicine, to senolytics.
I don’t think it’s a matter of if, only a matter of when. Ray Kurzweil’s prediction is the end of 2030. George Church and David Sinclair are slightly more conservative and put it in the mid-2030s.
Everybody’s responsibility today is to stay in the best health they can to intercept these technologies when they do materialize.
We would like to ask you a small favor. We are a non-profit foundation, and unlike some other organizations, we have no shareholders and no products to sell you. All our news and educational content is free for everyone to read, but it does mean that we rely on the help of people like you. Every contribution, no matter if it’s big or small, supports independent journalism and sustains our future.





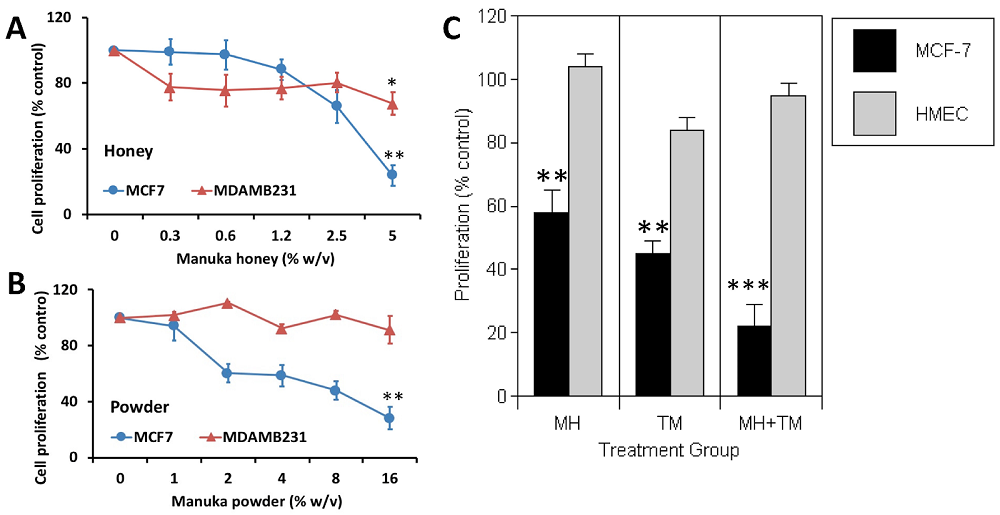
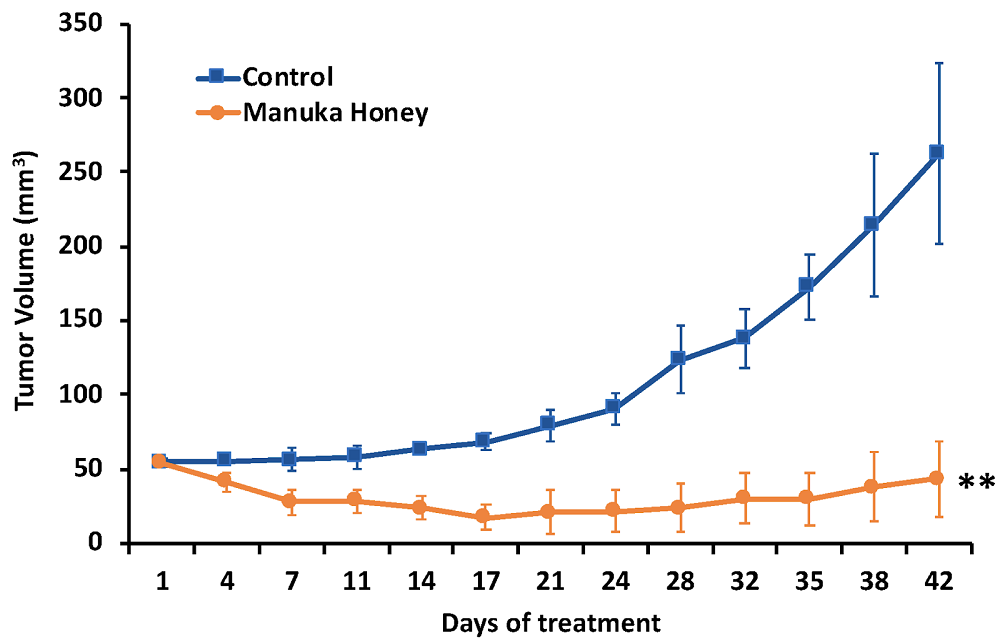
 Taken at the 4th TimePie Longevity Forum in 2024
Taken at the 4th TimePie Longevity Forum in 2024