After three years in digital format due to COVID concerns, one of the oldest longevity conferences was again welcoming attendants in New York City. Ending Age-Related Diseases (EARD), organized by lifespan.io for the sixth time, kept with modern ideas by using a hybrid format that combined in-person attendance with full online access in real time. This enabled the online participation of hundreds of experts and enthusiasts who could not travel to NYC in person.
This wasn’t the only aspect in which EARD-2023 led the way. The longevity space is changing, and we at lifespan.io are trying to be at the forefront of those changes. As we see it, one of the most profound shifts, although still in its early stages, has been the introduction of decentralized science, or DeSci.
DeSci provides a vibrant and exciting alternative to the traditional framework of scientific discovery and translation, improving incentives and removing roadblocks. A pioneer of crowdfunded science, lifespan.io has been very bullish about DeSci. We have collaborated with several DeSci organizations and initiatives, and so we have finally decided to bring DeSci to the main stage at EARD2023, officially naming it the Longevity + DeSci Summit.
While our previous conferences were mostly about science, this time, we fully embraced our advocacy side. There is a growing understanding in the field that advocacy is an integral part of the longevity ecosystem, indispensable for shifting public opinion and creating a favorable regulatory and investment climate.
We hosted our conference at Capitale, a historic building and the former seat of the Bowery Savings Bank, which retains a lot of its in-your-face grandeur. Turn-of-the-century interiors, complete with the bank vault and the lever-operated elevator, provided a refreshing contrast to the cutting-edge science being discussed on the stages and sidelines.

Inside the Capitale building
Unfortunately, we can only bring you a roundup of some of the talks from this extremely fast-paced and busy conference, which took place on two stages simultaneously, but videos will become available at some point.
Off to a good start
lifespan.io president Keith Comito commenced the conference with a fast-paced overview of trends in citizen and decentralized science. He began with the opening of the first citizen science laboratory in the world, Genspace, in NYC in 2010, and then continued with lifespan.io founding in 2014 along with our crowdfunding projects for SENS Research Foundation and other influential players in the longevity field. He noted how anti-aging research is being accelerated with novel tools and forms of organization, such as cryptocurrency, blockchain, and DAOs.
Keith made an important mention of earlier citizen initiatives, such as the Jimmy Fund, the massive grassroots campaign by Mary Lasker and Sidney Farber that elevated cancer research to the forefront of the US public agenda. We still have a lot to learn from those epic campaigns of old.
Keith then moved on to Zuzalu, the first-of-its-kind pop-up city that, earlier this year, brought together hundreds of people from several overlapping fields, including crypto, DeSci, and longevity, for two months of cohabitation and collaboration. Our team had boots on the ground in Zuzalu, and you can read all about it in our extensive coverage.
Briefly glancing over the improving attitudes towards longevity in society, Keith continued with an overview of lifespan.io and its history and contributions to the field.

History, however glorious, is still history, and Keith backed it up with plenty of exciting news to report. First, our collaboration with Chris Hemsworth’s fitness app and community Centr, which turned to us to provide millions of its users with quality longevity-related content. We are proud of this opportunity, which can serve as a blueprint for extending the reach of longevity advocacy.
Keith also reported on the creation of the first ever longevity-related philanthropy NFTs. Those NFTs, based on the ancient image of Ouroboros, the dragon-snake biting its own tail, are automatically enhanced when their owner donates to longevity-related causes, and are “demi-soulbound” – that is, they are still tradable, but their upgradable aspects are based on the donations of the current holder. This is yet another creative way to boost awareness and philanthropy in our field.

But creativity never sleeps. Keith announced yet another collaboration, this time with the game development company Skillcap Studios on a game based on the Fable of the Dragon-Tyrant by the philosopher Nick Bostrom. The fable allegorically depicts aging as a cruel dragon that had been demanding multitudes of sacrificial victims from people until they acquired enough strength and knowledge to revolt against him. According to Keith, the “play-and-earn gaming incentivizes players to be healthy and builds up donations to fund research which could literally save them in real life.” The game is expected in the second quarter of 2024.
Why DeSci?
Todd White, steward of VitaDAO, expertly argued a case for decentralized science. He recounted the well-known problems that “traditional” science has been mired in, such as publication bias.
Todd highlighted misaligned incentives, including the “publish or perish” paradigm, which leads to the kind of publication bias that pushes researchers towards a wide variety of conscious and subconscious actions that pursue a single goal: to get published whatever it takes.
In this system biased against risk, failure is not rewarded. However, the null hypothesis, Todd said, is not a failure devoid of value. It is an important piece of information that should be made available to the scientific community – which the current system often prevents.
The DeSci movement is well positioned to alleviate, if not solve, many of those problems by reimagining the system of incentives and making research more open and collaborative.
For instance, intellectual property stored as an NFT (IP-NFT), one of the pillars of DeSci, is not a gimmick but a useful feature that ensures all the information about a project is kept “on-chain”, verified and reviewed, and is an asset in itself. This de-risks the first steps in bringing scientific knowledge to the market.
Todd talked about The Longevist, a website launched by VitaDAO that publishes curated preprints. Preprints that have received the most votes from the community are highlighted. The curating team includes big names, such as Andrea Maier and Matt Kaeberlein. The Longevist positions itself as an alternative to scientific journals that act as “profit-hungry intermediaries”.
Todd also touched upon the topic of longevity advocacy. “The communication the value of longevity has not reached maturity yet,” he said. “When researchers try to explain the field, they think about nuances, but people want soundbites.” This echoes lifespan.io’s long-held views and relates well to our constant effort to hone our messages and the art of longevity advocacy as a whole.
Nature and longevity interventions work in different ways
One of the star guests of the conference was Vadim Gladyshev, a renowned Harvard geroscientist. In his talk, he started from the basics, by stating that there is still no agreement among the scientific community on what aging is. Different answers to this question, according to Vadim, lead to different strategies in fighting aging. Amazingly, with all the “geroscience boom” going on, there is not a lot of research into this fundamental question. “We need to agree on what we are optimizing”, Vadim said, “less chances of dying, fewer diseases, better function, or something else?”
While we can operate on all those levels, it is important to address the aging processes themselves. Vadim’s team developed a research program in order to address them directly. One of its parts attempts to understand what gives longevity to various systems, be it a cell type or an animal.
The second pillar is longevity interventions. While we know that some of them significantly extend lifespan in animal models, the question is if longevity of cells, longevity of organisms, and longevity-extending effects of certain interventions work along the same lines.
The third component is rejuvenation, the idea of reducing the biological age of an organism. It is different from aging, since a change from young to old is not necessarily the change from old to young, only in reverse. Questions of rejuvenation hence must be addressed separately. Working in all three directions will allow scientists both to slow and to reverse age-related changes.
Studying long-lived animals, Vadim’s team has discovered that the pathways leading to their exceptional longevity can differ between species. In a recent paper published in Cell, the group analyzed 41 species of mammals at an organ level to identify longevity signatures: omics-based patterns that illustrate the potential to live longer across mammalian species. The known maximum lifespan of a species is strongly correlated with patterns in gene expression, which provides a pathway towards longevity interventions.
Interestingly, “longevity signatures” across species do not correlate with the effects on gene expression produced by interventions. This means that the best interventions we have today, such as caloric restriction or rapamycin, work in different ways than those found by nature to extend maximum lifespan in long-lived species. This vast potential (after all, the difference in maximum lifespan across mammalian species is 100-fold) is “completely untapped”, according to Vadim, and longevity signatures provide “a direct tool for developing new interventions.”
It’s time to talk about female reproductive aging
After becoming a contributor to VitaDAO, Laura Minquini thought the model could be used to promote a highly important yet unfairly overlooked part of the longevity field – female reproductive aging. To turn things around, Laura created AthenaDAO, “a decentralized community to fund, govern, incubate, and support translational research into women’s health.”
Reproductive aging remains vastly under-researched, despite being relevant to half of the world’s population and to organismal aging in general. Beyond oncology, just 1% of healthcare research and innovation budget is spent on female-specific conditions. According to Laura, in longevity biotech investments, reproductive longevity comes second last, just before pets.
Decentralizing research into female reproductive longevity, Laura said, will empower women to invest directly into the topic that is highly relevant to them, circumventing the traditional avenues that apparently de-prioritize their plight. However, it is not only about research but also about education, advocacy, and access to resources. The aspiration, according to Laura, is to create a global female-led movement.
Formed only a year ago, in August 2022, AthenaDAO had already issued one IP-NFT to fund a research project on diminishing ovarian reserves. Six research projects are under advanced review as well as one stealth company. AthenaDAO prides itself on being the second biggest bio-DAO after VitaDAO.
Laura’s pitch to biotech is “Give women what they need, and they’ll become your best customers”. The menopause market, she said, is evaluated at 600 billion dollars, and yet research into menopause and its effects on the female organism is scarce. For instance, there are no biomarkers for when menopause begins (premenopause).
From the conference’s stage, Laura addressed the scientific community with a call for collaboration in several areas such as mitochondrial dysfunction, inflammaging, and the role of extracellular matrix in uterine pathologies.
lifespan.io – an ecosystem-creating community hub for the field
The second day of the conference started with a keynote talk from lifespan.io executive director Stephanie Dainow. “There are a lot of longevity conferences going on,” she said. “What we tried to be exceptional at is expanding and diversifying our audience. We have people from many different communities here: crypto, DeSci, government, and even general public, because we need to build the community.” Here are some excerpts from Stephanie’s talk:
Whether you are a transhumanist who wants to upload their mind, it doesn’t matter, because the first step we must take is to understand aging, and to do that, we need more funding, knowledge, collaboration, we need a united voice, so we can operate together more effectively.
To amplify the great work the scientific community is doing, we need precision and thoughtfulness in our messages, which lifespan.io embodies. For a decade, we have been known for our unwavering dedication to responsible and high-quality longevity-related content, and our deep engagement with diverse audiences. We now have hundreds of thousands of monthly visitors to our website, and millions of subscribers across our various YouTube channels. We are a source of education and inspiration. We are bridging the gap between the research and the public, having become one of the foremost non-profits in the longevity domain.
We are architects of connection, introducing organizations and individuals to each other and actively supporting collaborations. The recent rapid expansion of the longevity field generated a clear need for a roadmap. We are guardians of progress, equipping scientists, investors, policy makers, and all community members with the tools they need to navigate this complex landscape. Our purpose is to facilitate cross-pollination, sparking those invaluable serendipitous connections. An embodiment of this is our Longevity Investors Network.
We have not yet reached the level of impact that was characteristic of the campaigns of old that fueled research into cancer and Alzheimer’s. In another example, climate change, once relegated to scholarly circles, now commands worldwide attention. Irrespective of differing viewpoints, we cannot deny the remarkable impact achieved. We need to recreate this success in our field.
Every interaction is an opportunity to kindle a flame, to attract fresh champions to our mission. Yet, the message must be meticulously crafted. We at lifespan.io possess the expertise to shape the discussion, to counter the misconceptions. We tread this delicate line between exciting and overpromising. Collaborative problem solving, tolerance towards each other’s views, deduplication of efforts and effective resource allocation – lifespan.io is a hub that supports all of this.
Making old drugs young again
Brian Kennedy, professor of biochemistry and physiology at the National University of Singapore and a star geroscientist, delivered a captivating update on his and his team’s work.
One of the problems with clinical trials in medicine is what Brian calls “the sickcare approach” which prioritizes treating symptoms of the disease after they appear over preventing them from appearing in the first place. In part, this stems from the Hippocratic Oath being reduced to one of its postulates, “do no harm”, which can be a road to hell paved with good intentions.
This entrenched approach makes it harder for the medical establishment to accept the need for healthy longevity studies. “We can’t work on someone who’s not sick”, many healthcare professionals would say. But doing nothing, Brian argued, is actually harming people. “This paradigm needs to be completely revisited,” he said. “Waiting until people get sick in their 50s and 60s is one of the least effective strategies you can take. You’re waiting till the problem is difficult before trying to solve it.”
Brian gave an overview of the studies his group is conducting on various potentially life-extending small molecules. Since even in mice, lifespan studies take a long time and are very expensive, the team has moved towards using biomarkers of aging and function in studies six to eight months long. This also allows for better histological analysis. “A complete change of strategy”, Brian called it.
The group pours additional resources into studying several doses rather than one arbitrarily chosen dose and making sure that both sexes are adequately represented, which is important given significant sex-related differences in aging (and, therefore, in the effects of anti-aging therapies).
Among the molecules currently being tested, Brian highlighted gemfibrozil, which reduced frailty in mice by 45%. Using a novel technique, the group was able to prove that previous research has misidentified gemfibrozil’s target as PPAR. The group has identified three true targets of gemfibrozil.
With another promising compound, urolithin A, the group saw a significant reduction of frailty in mice and “a very robust extension” in lifespan, although just in flies and killifish for now.
Many of the drugs Brian’s team is working on have been in use for decades, but they apparently can be repurposed as anti-aging drugs. Brian called this approach “making old drugs young again”.
Brian highlighted the importance of the combination approach in longevity medicine, and the fact that we are still not well-versed in creating such combinations. “When we combine things in mice,” he said, “we mostly get nothing – i.e., the effect size of the best single intervention. In many cases, the drugs actually cancel each other out. A cumulative effect is only the third most probable outcome”.
Mammals, unite!
At the beginning of his talk, Steve Horvath, “the father of methylation clocks” and a principal investigator at Altos Labs, outlined a major challenge in geroscience: the need for high quality data for AI-powered research and for full access to this data.
Thankfully, as a step in the right direction, the Mammalian Methylation Consortium founded by a group of scientists including Steve himself, just released data from more than 15,000 samples taken from 70 tissue types of 348 mammalian species. The data and the software to explore it are widely available, and in his talk, Steve called on anyone interested to put it to good use. Today, the Consortium includes more than 200 collaborators, an unprecedented number for our field.
According to Steve, a good biomarker of mammalian aging should be applicable to all mammals, because, in his opinion, all mammalian species age, even those who are sometimes credited with “negligeable senescence”.Steve’s group has even been able to develop an epigenetic clock for the naked mole rat. Most of Steve’s talk was dedicated to this particular statement.
To build this multi-species clock, the group had to work with tens of thousands of methylation sites that are highly conserved in mammals. This allowed them to build what Steve called the third-generation epigenetic clock.
Steve introduced the concept of relative age, which works like this: if the maximum lifespan in rats is four years, a two-year-old rat’s relative age would be 0.5, the same as the relative age of a 60-year-old human. Third-generation clocks predict this relative age based on methylation.
However, is it really possible to construct a clock that would be applicable to every mammalian species, without exception, given that mammals are separated by as many as 200 million years of evolution and their maximum lifespans differ 100-fold?
The answer, according to Steve, is yes, and the proof was recently published in Nature Aging. This new clock achieves remarkable correlation across all species, which, Steve said, shows that “something is deeply conserved about all those species, as if there is some clock ticking.” Here is how well this single clock applies to different mammalian species with vastly different maximum lifespans:

The new clock has been validated in vitro, showing an almost complete “wipeout” of epigenetic age following cellular reprogramming in human fibroblasts. Known anti-aging and pro-aging interventions (such as caloric restriction and high-fat diets, respectively) move the needle of the new clock in expected ways.
According to Steve, one of the inferences of this remarkable cross-species robustness is that if an intervention moves the needle in one mammalian species, it will probably do the same for other species, including humans.
Analyzing this pan-mammalian methylation data revealed yet another interesting finding: methylation signatures of popular anti-aging interventions do not match those associated with maximum lifespan. This suggests that while those interventions can make individuals live healthier and longer, they can hardly extend their lifespan beyond the maximum one for their species, which is consistent with a lot of previous research. To extend human maximum lifespan, new, currently undiscovered interventions are probably needed.
Can it even be done? Steve said that maximum lifespan is strongly correlated with the rate of change in methylation. Probably, anything that drastically slows this rate of change in humans has the potential to affect our maximum lifespan.
Finally, Steve reported that across multiple mammalian species, the rate of change in methylation is higher in young animals than in old ones. This interesting finding suggests that methylation is linked to development: starting from the moment of the embryonic reset, we basically begin to age rapidly, even though it will take decades for aging to manifest itself as dysfunction.
The two types of aging
In an interesting counterpoint to Steve Horvath’s talk, Peter Fedichev, CEO of Gero, gave a talk with the inspiring title of “How to Stop Worrying and Halt Aging”. However, the contents of the talk might have been less optimistic than the title.
A while ago, Peter, a physicist-turned-geroscientist, got the longevity field’s attention with his theory of aging, which postulates that humans and mice age fundamentally differently, even though mice are the most popular animal model in geroscience. In mice, dysregulation starts creating damage early on, and this damage becomes a runaway train, leading to a short maximum lifespan. In humans, remarkably long-lived species, potent regulatory mechanisms maintain homeostasis for a long time, but when they themselves become dysregulated, this is the point when we start experiencing accelerated aging.
Two major upshots from this theory are that mice are probably a bad animal model for discovering anti-aging interventions for humans and that slowing or even stopping aging in humans is fundamentally easier than reversing it (“true rejuvenation”). Peter’s theory, while far from being confirmed or even widely accepted, is interesting and worth keeping in mind. Read our recent interview with Peter, where it is explained in much more detail.
Is a super-drug coming?
Maxwell Biosciences, although not a household name in the longevity field, might be one of the most interesting companies in it. J. Scotch McСlure, its CEO, has a lot of experience and a fascinating personal history, as he got into biotech to save his father and daughter from disease. His company’s idea is to create a “synthetic immune system”.
Maxwell is currently working on a molecule with a wide variety of effects: it is effective against viruses, fungi, and bacteria, and it also has anti-cancer properties. Maxwell was initially funded by DARPA to create a “synthetic immune system”. It started with the peptide LL-37, which was discovered in heterochronic parabiosis studies. According to McClure, this unassuming molecule serves as an “orchestra conductor” for the entire immune system by recruiting immune cells and facilitating tissue repair and cleaning. Maxwell has been able to create a molecule that mimics LL-37 and is currently taking it to clinical trials.
Maxwell has received both government and private funding and conducted extensive preclinical studies that confirmed the molecule’s effectiveness against all tested enveloped viruses, drug resistant fungi, and more than 70 bacterial strains. Maxwell is collaborating with the Barron Lab in Stanford and Kirshenbaum Lab NYU, which pioneered this research about two decades ago.
LL-37 is a part of the “healing secretome”, as it is secreted by many tissues and universally present across the body. However, in vivo, it breaks down unpredictably and quickly, which precludes its use as a drug. The solution found was quite ingenious: to replace the carbon-based backbone of the peptide, vulnerable to degradation by protease enzymes, with a nitrogen-based one while leaving all the side chains intact. This modification also reduced the molecule’s size to the point that it is regulated by the FDA as a small molecule.
Maxwell has chosen infectious diseases as its indication, which allows for a much quicker and cheaper FDA approval process compared to fields like cardiovascular diseases or cancer. However, their discovery is potentially highly relevant to the aging processes, and we will probably be hearing about Maxwell a lot in the coming years.
We would like to thank our wonderful sponsors:
Platinum
Date Posted: August 28, 2023
Researchers publishing in Journal of the American Veterinary Medical Association have conducted a feline clinical trial finding that rapamycin is effective against heart enlargement in cats.
A well-known drug for a well-known problem
Rapamycin, also known as sirolimus, is one of the most well-known and commonly taken compounds for longevity. The key metabolic factor it inhibits, the mechanistic target of rapamycin (mTOR), has two protein complexes, mTORC1 and mTORC2, both of which have substantial downstream effects, including in the cardiovascular system [1]. In this and other organs, inhibiting mTORC1 has positive effects, while inhibiting mTORC2 has negative ones. However, rapamycin affects mTORC1 much more acutely and mTORC2 only over the long term, meaning that intermittent dosing can be an effective strategy [2].
Hypertrophic cardiomyopathy (HCM), the unhealthy enlargement of the walls of the heart, affects approximately 15% of domestic cats and is often found not to have any clear cause [3]. This disease is largely untreatable by downstream therapies and, just like in people, it often leads to heart failure and death. As rapamycin was found to be effective in a mouse model of cardiac hypertrophy [4] and another study found it to improve the hearts of dogs [5], these researchers decided to see if it was effective in cats as well.
Conducted like a human clinical trial
As this study was conducted on companion animals recruited from the general population, it was conducted much like a human trial. This study was fully blinded, and the owners signed informed consent forms. One-third of the enrolled cats received placebo, one-third received low-dose delayed-release rapamycin (0.3 mg/kg), and the last third received high-dose delayed-release rapamycin (0.6 mg/kg).
Attempting to treat HCM before it became a problem, this study focused on subclinical HCM with no other diagnoses; cats with severe health problems were excluded from the study, as were cats over the age of 12 (in human years, approximately retirement age). These cats were 6 years old, with a wide variety of ages. 53 cats were screened, and 36 of them finished this 180-day trial.
While a substantial number of adverse events appeared in this study, they applied to all three groups mostly equally. Although one cat in the high-dose group died of diabetic ketoacidosis, rapamycin was not found to be a statistically significant cause of any adverse event. Rapamycin was also not found to cause changes in body weight, heart rate, systolic blood pressure, quality of life, or biochemical measurements.
Not all of these groups started with the same maximal wall thickness (MWT). The placebo group, on average, had thicker walls than the treatment groups. After 60 days, these groups did not change much, with the low-dose group slightly thinning their walls. However, after 180 days, the placebo group’s walls thickened substantially, which did not occur in either of the rapamycin groups.

The researchers also found a biomarker for the development of feline HCM. Cats with more N-terminal pro-B-type natriuretic peptide (NTproBNP) at baseline were substantially more likely to develop thicker walls. Rapamycin seemed to blunt this correlation and reduce NTproBNP, although these differences did not reach the level of statistical significance.
Finding the right dose
With the effects of mTORC1 and mTORC2 in mind, these researchers believe that low-dose delayed-release rapamycin appears to be more effective and potentially safer than its high-dose counterpart, at least in cats suffering from HCM.
This study had some limitations, including the relatively small study groups and the lack of direct measurements of mTORC1 and mTORC2. This study also focused on rapamycin itself rather than rapalogs that only affect mTORC1. The researchers note that further studies would be required to determine if their approach has an appreciable effect on lifespan and on HCM over the long term.
We would like to ask you a small favor. We are a non-profit foundation, and unlike some other organizations, we have no shareholders and no products to sell you. All our news and educational content is free for everyone to read, but it does mean that we rely on the help of people like you. Every contribution, no matter if it’s big or small, supports independent journalism and sustains our future.
Literature
[1] Sciarretta, S., Forte, M., Frati, G., & Sadoshima, J. (2018). New insights into the role of mTOR signaling in the cardiovascular system. Circulation research, 122(3), 489-505.
[2] Arriola Apelo, S. I., Neuman, J. C., Baar, E. L., Syed, F. A., Cummings, N. E., Brar, H. K., … & Lamming, D. W. (2016). Alternative rapamycin treatment regimens mitigate the impact of rapamycin on glucose homeostasis and the immune system. Aging cell, 15(1), 28-38.
[3] Kittleson, M. D., & Côté, E. (2021). The feline cardiomyopathies: 2. Hypertrophic cardiomyopathy. Journal of feline medicine and surgery, 23(11), 1028-1051.
[4] Shioi, T., McMullen, J. R., Tarnavski, O., Converso, K., Sherwood, M. C., Manning, W. J., & Izumo, S. (2003). Rapamycin attenuates load-induced cardiac hypertrophy in mice. Circulation, 107(12), 1664-1670.
[5] Urfer, S. R., Kaeberlein, T. L., Mailheau, S., Bergman, P. J., Creevy, K. E., Promislow, D. E., & Kaeberlein, M. (2017). A randomized controlled trial to establish effects of short-term rapamycin treatment in 24 middle-aged companion dogs. Geroscience, 39, 117-127.
Date Posted: August 28, 2023
“Could humans become immortal?” is something we get asked quite often, and the answer depends on what exactly you mean.
When it comes to immortality, what you mean is important
Whether human immortality is possible greatly depends on how you define it. If you define it as living forever and being unkillable like in a comic book or movie, then, no, it is highly unlikely.
However, if you define it in terms of showing no decline in survival characteristics, no increase in disease incidence, and no increase in mortality with advancing age, then yes.
To some people this may seem to be a matter of semantics, but it is not. The first is a science-fiction fantasy; the second is based on real-world biology that evolution has already selected for in certain species. This is known as negligible senescence. And in fact, some animals are already doing exactly this!
And the good news is that there is no reason why humans could not enjoy considerably increased healthy longevity if the appropriate technologies are developed. It isn’t even beyond the realms of possibility that humans might achieve negligible senescence thanks to the march of medical science and technology.
Senescence and negligible senescence
Senescence refers to the gradual deterioration of aging and is typically very obvious in almost every species. More accurately, senescence refers to a decline of survival characteristics, such as strength, mobility, and senses, and age-related increases in mortality along with a decrease in reproductive capability. Mortality rates for humans and most animals increase dramatically with age beyond reaching reproductive maturity.
A number of species age very slowly and enjoy extreme longevity and a few species are even more unusual and exhibit negligible senescence. An organism is considered negligibly senescent (NS) if it does not show any loss of survival characteristics, such as strength, mobility, and senses, an increased mortality rate with advancing age, or a loss of reproductive capability with age.
| Species |
Recorded lifespan
|
| Rougheye rockfish |
205 years[1-2] |
| Aldabra Giant Tortoise |
255 years |
| Lobsters |
100+ years (Presumed NS) |
| Naked mole rat (Heterocephalus glaberis) |
28 years |
| Sea anemones |
60–80 years |
| Freshwater pearl mussel |
210–250 years[3] |
| Ocean Quahog clam |
507 years[4] |
| Greenland Shark |
400 years |
| Lake sturgeon (Acipenser fulvescens) |
152 years (Presumed NS) |
| Clams such as Panopea generosa |
160 years (Presumed NS) |
Possibly even more intriguing is the hydra, a species that is observed to have no lifespan limit, as it regenerates very quickly. Barring predation and changes to its environment, it is one of the few species for which the phrase “biological immortality” would be appropriate [5]. The hydra is quite unique in how its cells work, and it is quite unlike the majority of other organisms on the planet; it is a true oddball but fascinating all the same.
Can we achieve negligible senescence?
Well, this is great news if you happen to be a lobster and avoid the fisherman’s pot long enough to reach a ripe old age, but what about us; how can we benefit from the same advantages that negligibly senescent species do?
It is clear that we would have to wait a long time, perhaps forever, before evolution selected the same traits in humans, so something a little more direct is needed.
The good news is that some researchers are trying to find out if the longevity of the naked mole rat might be exported to humans. In fact recently scientists successfully transfered a longevity mechanism from naked mole rats to mice [6]. This opens the door for potentially doing the same for humans. Who knows, we might be able to harness the superior DNA repair of the naked mole rat and signficantly reduce the impact of aging on our bodies.
Some scientists, such as Dr. Aubrey de Grey, propose that we can engineer negligible senescence by using a repair-based approach to the damage that aging causes. This is the basis of SENS, the strategies for engineered negligible senescence, and is being pursued by the SENS Research Foundation.
Other researchers have built on the original concept of SENS, and in 2013, the Hallmarks of aging was published. This landmark paper broke the aging process down into nine distinct processes known as hallmarks and essentially gave researchers a way to classify aging and an insight into what processes they might target to slow down or even reverse aging.
While there are quite a number of aging theories, the Hallmarks of aging appears to be one of the more popular, judging by how many times it has been cited and how often it is used in academia. Essentially, the Hallmarks of aging has given researchers a list of targets to develop therapies for and now the race is on to create them.
Should negligible senescence be achieved in humans through SENS or other approaches such as partial cellular reprogramming, it would potentially mean the end of age-related diseases and ill health, a most worthy goal indeed.
Negligible senescence does not mean they cannot die
It is worth noting that even though a number of species enjoy negligible senescence and do not age or age immeasurably slowly, they are still vulnerable to predation, accidents, starvation, environmental dangers, changes to their environmental niches, and diseases. This means that extremely old examples of these species with negligible senescence are very rare, especially in the wild.
To further complicate matters, we often need to sacrifice the animal in order to measure its age by examining the deep tissues and marks inside bones, much like measuring rings in a tree trunk.
This means that we cannot know the maximum age that might be achieved by these species, so the above numbers are based on what information we have; there could well be considerably older examples out there.
The point here is that negligible species with senescence do not deteriorate with age and may live considerably longer than has been recorded.
The inevitability of multicellular aging
Back in 2017, a great deal of fuss was made about humans achieving negligible senescence, with a number of articles suggesting that it is impossible. The reason is that the mainstream media interpreted a research paper very badly, assuming that the authors imply that because aging is inevitable, we cannot do anything about it [7].
The media was filled with articles smugly proclaiming in some cases that aging is unstoppable and mathematically impossible to defeat. The problem with this interpretation is that it is just plain wrong. The original paper is, strictly speaking, correct in that aging damage is indeed inevitable, but it makes no assumptions about interventions. The publication says a great deal about what evolution has done and is likely to do based on observation, but that says absolutely nothing about what medicine may achieve in the future.
One cannot apply such thinking when it comes to engineering negligible senescence in humans through the periodic repair of age-related damage. So, quite simply, publications like this make little difference to work in this field, and they change the plausibility of us achieving negligible senescence in no way whatsoever.
No, aging is not inevitable
A more recent example in 2021, which was again met with almost gleeful declaration by the press that aging cannot be stopped, was the ‘invariant rate of ageing’ paper [8]. Unfortunately, once again the reporting was based on the a similar misunderstanding of what the study actually said.
The study was actually not a study about longevity or that aging was inevitable. It was trying to understand what influences the rate of aging across species and how much results from evolved biological processes versus the effects of the environment. While the research itself has obvious merit scientifically speaking, the popular press chose to represent it in a different light.
The irony is that instead of showing that aging is indeed inevitable, the research instead shows that eventually humanity will run out of ways in which environmental improvements will increase our lifespans. At that point further gains will only be achieved through medical interventions that address the aging processes directly and either repair the damage aging does, or slow aging down by make us more resilient.
There is a clear difference between Hollywood-style immortality and negligible senescence, with the latter being a plausible goal in the next few decades. Evolution has already demonstrated that negligible senescence is indeed possible; now, the next big challenge is to use an engineering approach to aging to see if we can emulate in people what nature has done in a few lucky species. The good news is, we have a list of targets and a far better understanding of what aging is than we did even 10 years ago, and there is a lot more interest in tackling aging from both the academic and investment communities.
Literature
[1] Munk, K. M. (2001). Maximum ages of groundfishes in waters off Alaska and British Columbia and considerations of age determination. Alaska Fish. Res. Bull, 8(1), 12-21.
[2] Cailliet, G. M., Andrews, A. H., Burton, E. J., Watters, D. L., Kline, D. E., & Ferry-Graham, L. A. (2001). Age determination and validation studies of marine fishes: do deep-dwellers live longer?. Experimental gerontology, 36(4), 739-764.
[3] Ziuganov, V., Miguel, E. S., Neves, R. J., Longa, A., Fernández, C., Amaro, R., … & Johnson, T. (2000). Life span variation of the freshwater pearl shell: a model species for testing longevity mechanisms in animals. AMBIO: A Journal of the Human Environment, 29(2), 102-105.
[4] Munro, D., & Blier, P. U. (2012). The extreme longevity of Arctica islandica is associated with increased peroxidation resistance in mitochondrial membranes. Aging cell, 11(5), 845-855.
[5] Martı́nez, D. E. (1998). Mortality patterns suggest lack of senescence in hydra. Experimental gerontology, 33(3), 217-225.
[6] Zhang, Z., Tian, X., Lu, J. Y., Boit, K., Ablaeva, J., Zakusilo, F. T., Emmrich, S., Firsanov, D., Rydkina, E., Biashad, S. A., Lu, Q., Tyshkovskiy, A., Gladyshev, V. N., Horvath, S., Seluanov, A., & Gorbunova, V. (2023).
Increased hyaluronan by naked mole-rat Has2 improves healthspan in mice.
Nature, 10.1038/s41586-023-06463-0. Advance online publication. https://doi.org/10.1038/s41586-023-06463-0
[8] Nelson, P., & Masel, J. (2017). Intercellular competition and the inevitability of multicellular aging. Proceedings of the National Academy of Sciences, 201618854.
[8] Colchero, F. et al. The long lives of primates and the ‘invariant rate of ageing’ hypothesis. Nature Communications (2021), doi: 10.1038/s41467-021-23894-3
Date Posted: August 25, 2023
A new study suggests that greater diversity of everyday activities is associated with a slower rate of age-related cognitive decline, independently of the amount of physical activity [1].
Variety or quantity?
It is widely accepted that being cognitively and socially active can slow down age-associated cognitive decline. Scientists also generally agree that the range of activities matters as well: the more you diversify them, the slower the decline [2]. Varied daily activities make people switch between tasks and adapt to new settings, which, as one study found, is likely to stimulate the hippocampus [3], a crucial brain area responsible for navigation, learning, and memory. Another study found a link between activity variety and actual hippocampal volume [2].
However, what if the effect is explained by more diverse activities being more physically demanding? After all, greater physical activity is also associated with slower cognitive decline [4]. Whether better physical shape produces the diversity of activities, or vice versa, it is possible that the relationship that is observed with cognitive function is driven by the level of physical activity rather than by the diversity of cognitive and social activities.
Diversity seems to matter
In this new study, the researchers attempted to answer this question by studying 252 community-dwelling (that is, not living in an institution) elderly adults with a mean age of 74 years. The participants completed a battery of cognitive tests at baseline and then self-reported their participation in ten common types of activity, such as reading, chores, and social visits, every three hours for five or six days. At the same time, the actual levels of their physical activity were recorded by wearable accelerometers. Results were controlled for age, sex, racial/ethnic minority status, marital status, education, self-rated health, depressive symptoms, and, interestingly, extroversion.
The researchers provide an example of how the ten daily activities they monitored differed for two of the study’s participants:

The main upshot is that greater diversity in daily activities was indeed associated with better overall cognitive functioning. Moreover, the effect size was similar to that of education, which, although possibly mediated by income, is a potent predictor of the rate of cognitive decline [5]. Interestingly, movement duration and step count did not show association with cognitive function.
Insights into the life of the elderly
The study also provided some interesting insights into the daily activities of the elderly. For example, chores and using computer or other electronic devices were the most common activities, with volunteering, work, and religious activities being the least common.
Average daily activity diversity did not differ between men and women, and it was greater among participants who were married or partnered. Importantly, activity diversity was substantially greater among non-Hispanic Whites compared to racial/ethnic minorities, despite movement duration and step count being mostly similar between the two groups.
This study features an interesting and compelling design but also has some serious limitations. For example, the authors did not control for income, which is known to be highly correlated with age-related cognitive decline and could at least partially explain the difference between minorities and non-minorities in this study.
While the study found no correlation between levels of physical activity and cognitive decline, the average daily step count was unusually low, which might have affected the results. Finally, it would be interesting to see longitudinal measurements of cognitive function. With the advance of commercially available wearable devices, setting up such studies should become increasingly easier.
Humans need to stay both mentally and physically active. DeYoung (2015) further asserts that the need to explore and adapt is fundamental for the health of any complex organism. The present study provides clarity to the complex question of how to best stay mentally active. People must constantly adapt to their world, even when engaged in routine activities that provide structure and meaning to daily life. The present study suggests that even daily routine activities, if combined with each other in a rich and balanced schedule, are related to higher levels of cognitive functioning that cannot be explained by the physical activity that accompanies these activities.
We would like to ask you a small favor. We are a non-profit foundation, and unlike some other organizations, we have no shareholders and no products to sell you. All our news and educational content is free for everyone to read, but it does mean that we rely on the help of people like you. Every contribution, no matter if it’s big or small, supports independent journalism and sustains our future.
Literature
[1] Brown, C. J., Jeon, S., Ng, Y. T., Lee, S., Fingerman, K. L., & Charles, S. T. (2023). Switching it up: Activity diversity and cognitive functioning in later life.Psychology and Aging. Advance online publication.
[2] Urban-Wojcik, E. J., Lee, S., Grupe, D. W., Quinlan, L., Gresham, L., Hammond, A., … & Schaefer, S. M. (2022). Diversity of daily activities is associated with greater hippocampal volume. Cognitive, affective, & behavioral neuroscience, 1-13.
[3] Aronov, D., Nevers, R., & Tank, D. W. (2017). Mapping of a non-spatial dimension by the hippocampal–entorhinal circuit. Nature, 543(7647), 719-722.
[4] Sofi, F., Valecchi, D., Bacci, D., Abbate, R., Gensini, G. F., Casini, A., & Macchi, C. (2011). Physical activity and risk of cognitive decline: a meta‐analysis of prospective studies. Journal of internal medicine, 269(1), 107-117.
[5] Zahodne, L. B., Stern, Y., & Manly, J. J. (2015). Differing effects of education on cognitive decline in diverse elders with low versus high educational attainment. Neuropsychology, 29(4), 649.
Date Posted: August 24, 2023
Publishing in Nature, a team of researchers including Vadim Gladyshev, Steve Horvath, and Vera Gorbunova has investigated the role of hyaluronan, which naked mole rats have in abundance, as a protective mechanism in a mouse model.
An established mechanism
Naked mole rats are notably resistant to cancer, and these researchers have previously determined that this resistance is, at least in part, due to high-molecular-mass hyaluronan (HMM-HA) [1]. Hyaluronan is a component of the extracellular matrix that is composed of long repeating chains of glucosamine and glucuronic acid. While smaller forms of hyaluronan (LMM-HA) are associated with inflammation [2] and cancer [3], HMM-HA is anti-inflammatory in nature [4], which may also explain some of the naked mole rat’s longevity.
However, while these benefits had been previously explored, the naked mole rat’s particular tissue-infusing amounts of hyaluronan had not been examined in other species. To that end, these researchers decided to create a mouse model that expresses HMM-HA in the way that the naked mole rat does. They used relatively large samples of mice for this lifespan study, with approximately 80-90 mice in both the treatment and control groups listed in the various results.
Not good for early development, but good for longevity
Because HMM-HA interferes with embryonic development and naturally accumulates in naked mole rats later in life, the researchers controlled its expression, activating it only upon injections of the commonly used molecule tamoxifen. Interestingly, the altered mice only had more HMM-HA in certain tissues when tamoxifen was administered; the muscle tissue contained eight times as much, the heart was unaffected, and the kidneys and intestines contained twice as much. The researchers note that the mice’s levels of hyaluronidase, which breaks down hyaluronan, were not affected and that naked mole rats have much less hyaluronidase.
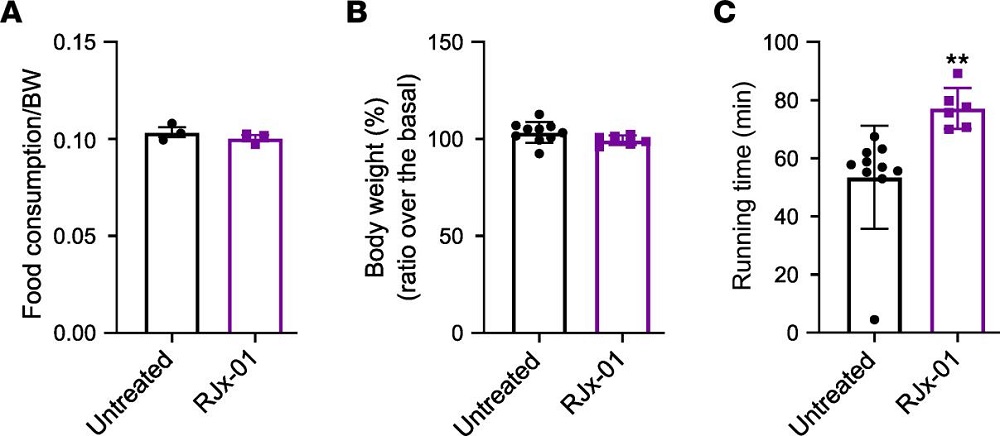
Even with that being the case, the mice were profoundly affected. Mice naturally get, and die of, cancer. 80% of the old, untreated mice had signs of cancer, but only 40% of the older treated group had such signs. Even when skin cancer was directly induced in the mice through application of a toxin, the HMM-HA mice had significantly fewer tumors.
With no change in overall bodyweight, there were multiple beneficial physical effects. Treated older mice performed better on the commonly used rotarod and forelimb grip strength tests. The treatment group retained stronger bone connectivity, was considerably less frail in old age, and was even biologically younger according to epigenetic testing done on liver tissue. Lifespan, itself, was significantly but slightly improved as a result of the HMM-HA treatment.

Measurements of gene expression and inflammation backed up these results. The treated group had fewer changes with aging and significantly less inflammation than the control group. The researchers note that these differences do not correspond to any other longevity treatment, suggesting that a combination treatment may be even more effective. Oxidative stress and intestinal health were also positively affected by the additional HMM-HA.
Already in use
Hyaluronan injections are already in use in the clinic for the treatment of arthritis. However, the hyaluronan in those treatments is not being produced by the body’s own cells and suffused throughout the muscles, as it was in these mice. Given the wide-ranging benefits displayed in this study, it may be worthwhile to determine if it is possible and safe to use mRNA or similar technologies to help human beings produce, and retain, more of this apparently life-extending compound.
We would like to ask you a small favor. We are a non-profit foundation, and unlike some other organizations, we have no shareholders and no products to sell you. All our news and educational content is free for everyone to read, but it does mean that we rely on the help of people like you. Every contribution, no matter if it’s big or small, supports independent journalism and sustains our future.
Literature
[1] Tian, X., Azpurua, J., Hine, C., Vaidya, A., Myakishev-Rempel, M., Ablaeva, J., … & Seluanov, A. (2013). High-molecular-mass hyaluronan mediates the cancer resistance of the naked mole rat. Nature, 499(7458), 346-349.
[2] Cyphert, J. M., Trempus, C. S., & Garantziotis, S. (2015). Size matters: molecular weight specificity of hyaluronan effects in cell biology. International journal of cell biology, 2015.
[3] Zhang, G., Lu, R., Wu, M., Liu, Y., He, Y., Xu, J., … & Gao, F. (2019). Colorectal cancer‐associated ~6kDa hyaluronan serves as a novel biomarker for cancer progression and metastasis. The FEBS Journal, 286(16), 3148-3163.
[4] Muto, J., Yamasaki, K., Taylor, K. R., & Gallo, R. L. (2009). Engagement of CD44 by hyaluronan suppresses TLR4 signaling and the septic response to LPS. Molecular immunology, 47(2-3), 449-456.