How Spaceflight Alters the Rate of Aging
A review published in Aging Cell has explored the impact of spaceflight on human and other organisms’ health and aging [1].
Hazards of spaceflight
Will sending humans to space make them age slower or faster? Will they be healthier, or will their health suffer? As space exploration progresses, researchers are working to find answers to those questions.
It is no surprise that NASA is investigating the impact of spaceflight on human health. Its Human Research Program has reported the five hazards of human spaceflight. Those hazards are space radiation, isolation, distance from Earth, altered gravity fields, and hostile and closed environments, all of which pose risks to human health. The most dangerous risks were categorized based on the systems they affect: visual, cardiovascular, central nervous, and musculoskeletal.
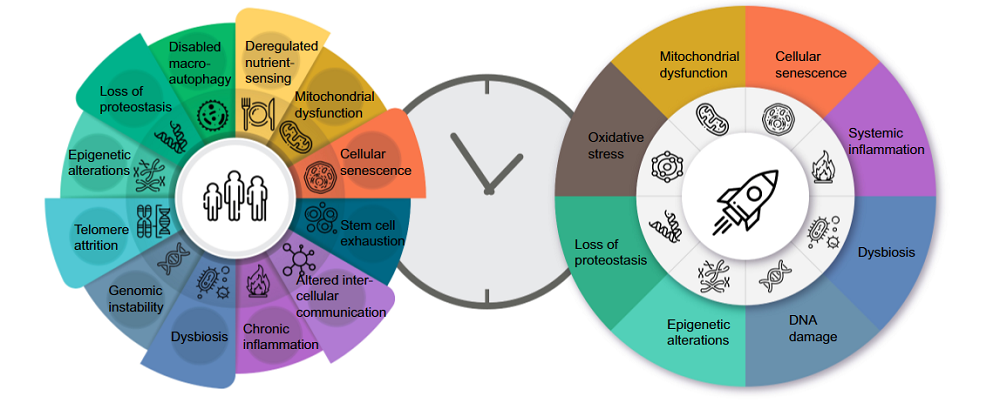
As shown below, previous research has summarized common features of aging on Earth and in space [2]. However, there is still a need to determine how long-term space missions would affect human health and, consequently, what the effect would be on aging.
Circadian rhythms in humans
Humans evolved to live on Earth. Our circadian rhythms reflect the Earth’s 24-hour light and dark cycles. Spaceflight disrupts those natural cycles; the sunrise-sunset cycle in Earth’s orbit lasts only 45 minutes.
Those circadian rhythms have an impact on health, including retinal function, cardiac metabolism, neural activity, and the formation of skeletal muscular tissue.
The circadian clock regulates some of the essential mechanisms of the cardiovascular system, such as blood pressure and heart rate [3, 4]. Research also suggests that an altered clock-associated mechanism is a risk factor for cardiovascular disease.
Additionally, circadian dysfunction occurs frequently in some aging-related neurodegenerative diseases, such as Alzheimer’s and Parkinson’s diseases [5]. Disruptions in circadian rhythms also result in such problems as sleep disruptions, sleep fragmentations, and reduced daytime alertness [6].
Scientists have been aware of space’s effect on circadian rhythms for over three decades. The molecules that control those cycles are called core-clock genes. Research has shown that in mammals on Earth, nearly 50% of expressed genes show rhythmic ~24-hour changes in expression in at least one organ [7].
There are also studies in fruit flies and rodents that were taken on space missions showing that clock genes play a significant role in pathologies associated with spaceflight. However, there is still a lack of thorough understanding of the molecular mechanisms of these conditions in humans.
Effects on aging and health
Some dysfunctions accelerate when people are in space. Changes in cardiovascular and musculoskeletal systems can be accelerated even 10-fold in space compared to on Earth [8].
Some of these changes are due to the lack of Earth gravity, such as muscle atrophy caused by muscle disuse. Astronauts can experience cephalad fluid shifts, which occur when fluids shift from the lower body toward the head and the heart. This results in smaller blood production, abnormal blood pressure, lower heart rate, and other cardiac issues. To prevent these changes from happening, astronauts need to follow strict exercise routines in space.
Astronauts also experience a condition known as Spaceflight-Associated Neuro-ocular Syndrome (SANS). SANS includes swelling of a portion of the optic nerve, retinal nerve fiber layer thickening, and other related conditions.
To compare organisms on Earth and during spaceflight, NASA performed a unique experiment. One of the astronauts sent for spaceflight had an identical twin. This way, researchers could monitor both siblings: one on a one-year space mission, the second on Earth.
Researchers observed multiple changes in the space-faring astronaut compared to the twin, including increased retinal thickness and changes in arterial dimensions. These changes reversed after six months upon return to Earth.
Researchers also observed a change in one of the Hallmarks of Aging: telomere length. While the astronaut was in space, researchers observed the astronaut’s telomeres lengthen, which suggests an anti-aging effect. However, 6 months after returning to Earth, the changes reversed. Moreover, following six months after the astronaut’s return to Earth, researchers observed shorter telomeres and more DNA damage, which suggests accelerated aging [9].
On the other hand, there are several reported anti-aging effects of spaceflight on humans and animals. A 520-day Space mission had an anti-aging effect on DNA methylation patterns in astronauts [10]. C. elegans worms and fruit flies in space showed slower aging [11, 12]. On the other hand, yeast fungi show signs of accelerated aging when exposed to simulated microgravity [13].
Longer and further space missions are planned for the future. As a part of the preparation for those missions, researchers need to determine in more detail how spaceflight influences the health and lifespan of astronauts.
Literature
[1] Malhan, D., Schoenrock, B., Yalçin, M., Blottner, D., & Relόgio, A. (2023). Circadian regulation in aging: Implications for spaceflight and life on earth. Aging cell, e13935. Advance online publication.
[2] Biolo, G., Heer, M., Narici, M., & Strollo, F. (2003). Microgravity as a model of ageing. Current opinion in clinical nutrition and metabolic care, 6(1), 31–40.
[3] Costello, H. M., & Gumz, M. L. (2021). Circadian Rhythm, Clock Genes, and Hypertension: Recent Advances in Hypertension. Hypertension (Dallas, Tex. : 1979), 78(5), 1185–1196.
[4] El Jamal, N., Lordan, R., Teegarden, S. L., Grosser, T., & FitzGerald, G. (2023). The Circadian Biology of Heart Failure. Circulation research, 132(2), 223–237.
[5] Leng, Y., Musiek, E. S., Hu, K., Cappuccio, F. P., & Yaffe, K. (2019). Association between circadian rhythms and neurodegenerative diseases. The Lancet. Neurology, 18(3), 307–318.
[6] Czeisler, C. A., Chiasera, A. J., & Duffy, J. F. (1991). Research on sleep, circadian rhythms and aging: applications to manned spaceflight. Experimental gerontology, 26(2-3), 217–232.
[7] Zhang, R., Lahens, N. F., Ballance, H. I., Hughes, M. E., & Hogenesch, J. B. (2014). A circadian gene expression atlas in mammals: implications for biology and medicine. Proceedings of the National Academy of Sciences of the United States of America, 111(45), 16219–16224.
[8] Vernikos, J., & Schneider, V. S. (2010). Space, gravity and the physiology of aging: parallel or convergent disciplines? A mini-review. Gerontology, 56(2), 157–166.
[9] Garrett-Bakelman, F. E., Darshi, M., Green, S. J., Gur, R. C., Lin, L., Macias, B. R., McKenna, M. J., Meydan, C., Mishra, T., Nasrini, J., Piening, B. D., Rizzardi, L. F., Sharma, K., Siamwala, J. H., Taylor, L., Vitaterna, M. H., Afkarian, M., Afshinnekoo, E., Ahadi, S., Ambati, A., … Turek, F. W. (2019). The NASA Twins Study: A multidimensional analysis of a year-long human spaceflight. Science (New York, N.Y.), 364(6436), eaau8650.
[10] Nwanaji-Enwerem, J. C., Nwanaji-Enwerem, U., Van Der Laan, L., Galazka, J. M., Redeker, N. S., & Cardenas, A. (2020). A Longitudinal Epigenetic Aging and Leukocyte Analysis of Simulated Space Travel: The Mars-500 Mission. Cell reports, 33(10), 108406.
[11] Honda, Y., Higashibata, A., Matsunaga, Y., Yonezawa, Y., Kawano, T., Higashitani, A., Kuriyama, K., Shimazu, T., Tanaka, M., Szewczyk, N. J., Ishioka, N., & Honda, S. (2012). Genes down-regulated in spaceflight are involved in the control of longevity in Caenorhabditis elegans. Scientific reports, 2, 487.
[12] Ma, L., Ma, J., & Xu, K. (2015). Effect of spaceflight on the circadian rhythm, lifespan and gene expression of Drosophila melanogaster. PloS one, 10(3), e0121600.
[13] Fukuda, A. P. M., Camandona, V. L., Francisco, K. J. M., Rios-Anjos, R. M., Lucio do Lago, C., & Ferreira-Junior, J. R. (2021). Simulated microgravity accelerates aging in Saccharomyces cerevisiae. Life sciences in space research, 28, 32–40.