Current Clinical Trials of Alzheimer’s Drugs
- More clinical trial results should be reported this year.

- The number of clinical trials targeting Alzheimer’s disease is increasing.
- Most of the new candidate drugs target neurotransmitters, immune processes, tau, and other components of Alzheimer’s instead of amyloid beta.
This month, a group of researchers published an annual report on the clinical trials that are testing drugs for Alzheimer’s disease. Overall, they reported an increase in the number of trials, with 158 drugs investigated across 192 trials [1].
A growing problem
The projections regarding the prevalence of Alzheimer’s disease are merciless. While in 2017, the number of people with detectable Alzheimer’s in the United States was estimated to be around 50 million, this number is projected to almost double by 2060. However, not all of these people will suffer from cognitive impairment, a symptom that most people associate with Alzheimer’s disease. These statistics and projections include people within the whole continuum of Alzheimer’s disease progression, including asymptomatic biomarker-positive people (46.7 million in 2017 versus a projected 75.68 million in 2060), people with mild cognitive impairment (MCI) with Alzheimer’s disease pathology (2.43 million vs. 5.7 million), and people with Alzheimer’s disease dementia (3.65 million vs. 9.3 million) [2]. These projections make the need for a treatment to address the diseases urgent.
The annual report
Over the past decade, the authors of this paper have been preparing an annual report on the state of clinical trials for Alzheimer’s disease drugs. This year, they reported that in the last decade, the number of trials and tested agents increased by approximately 35% and 40%, respectively, and in 2025, 59 new trials have been registered.
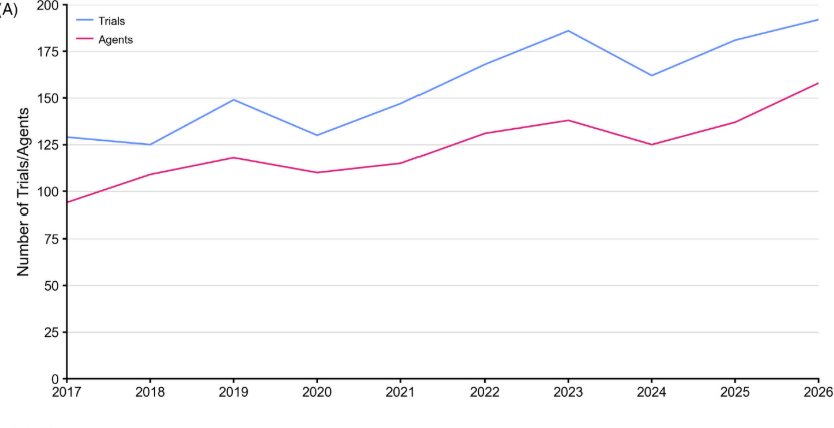
In this year’s publication, they reported on the state of the tested interventions as of January 1, 2026. Among the agents investigated, almost three-fourths are intended to slow disease progression, including small-molecule disease-targeting therapies (DTTs) (39%), which include drugs usually taken orally, and biologic disease-targeting therapies (34%), which include such treatments as monoclonal antibodies, vaccines, and antisense oligonucleotides. Other agents under investigation include cognition-enhancing symptom-targeted therapies (STTs) (18%) and drugs targeting neuropsychiatric symptoms (10%). Not all of the drugs are new; around 35% are repurposed drugs: drugs that have already been approved for other conditions and are now being tested for their effectiveness in Alzheimer’s disease.
Given the large potential market for Alzheimer’s disease drugs, it’s not surprising that the pharmaceutical industry sponsors 59% of all clinical trials.
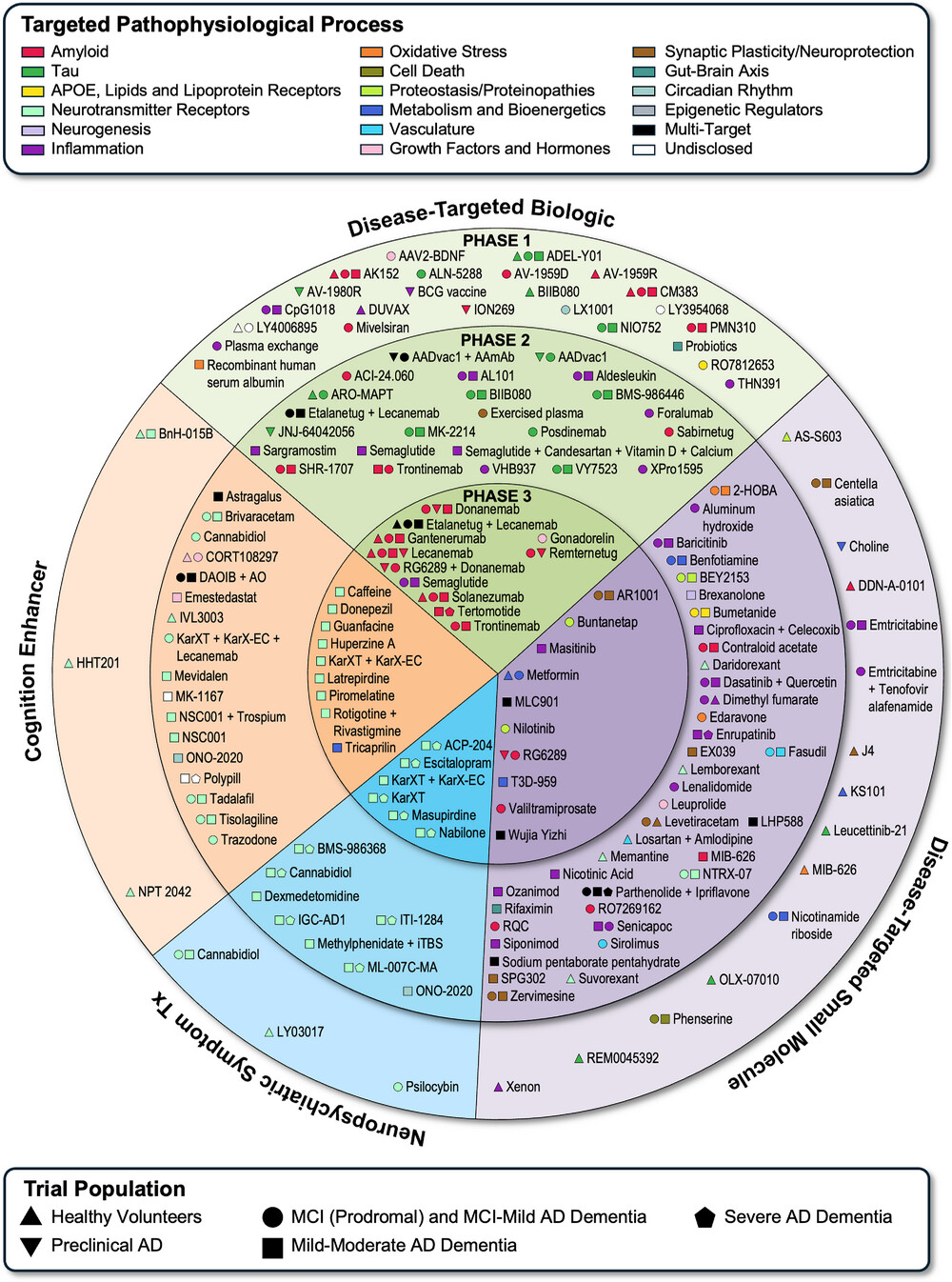
The drugs currently under investigation target diverse biological processes and are mostly tested as single agents (i.e., targeting a single process); however, several trials combine multiple agents. “It is clear that Alzheimer’s is a complex disease with many contributing elements,” said Jeffrey L. Cummings, M.D., ScD, from the Kirk Kerkorian School of Medicine at UNLV and the corresponding author of the study. “Inflammation is consistently present in the brain of Alzheimer’s patients, and reducing the inflammatory response promises to slow the disease process. Researchers are seeking ways to complement the approved anti-amyloid therapies. The development of combination therapies is likely, and combinations are being studied in current trials,” he said.
To map which biological processes are targeted by different trials, the authors used descriptive categories of the Common Alzheimer’s Disease Research Ontology (CADRO). Most commonly targeted CARDO categories include neurotransmitter receptors (24% of drugs), inflammation/immune processes (18%), Aß protein-related pathophysiology (16%), and tau-related processes (9%).
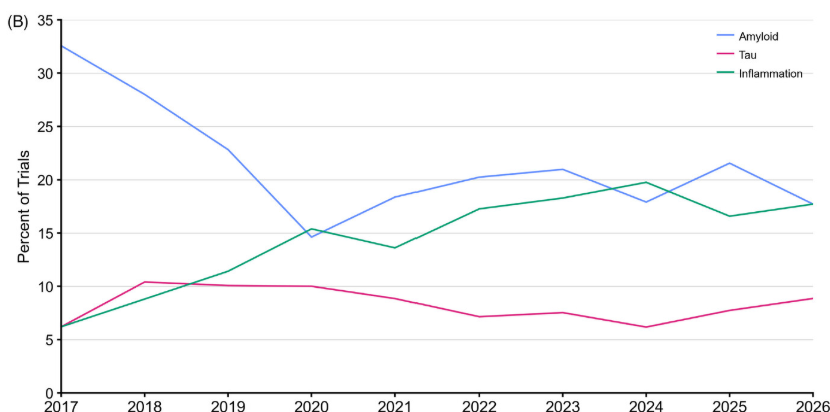
Over the past decade, there has also been a general shift in the Alzheimer’s disease-related molecular processes targeted in trials. Both inflammation and immune dysfunction, as well as tau-targeted agents, increased from around 6% to around 20% of the pipeline, while amyloid-targeted agents decreased from 33% to 20%.
The authors of the report point out that most clinical trials make use of Alzheimer’s disease biomarkers. Biomarkers are used “to confirm the diagnosis of Alzheimer’s disease for trial eligibility, stratify or enroll patients based on likelihood of progression, monitor therapeutic effects longitudinally, and verify the pharmacodynamic effect of the candidate therapy as a study outcome.“ [3]
The source of data
The data used for this report were collected by the authors for active trials as of January 1, 2026, from clinicaltrials.gov, “a clinical trial registry maintained by the US National Library of Medicine of the National Institutes of Health (NIH).” The trials that are required to be registered on that website “have at least one site in the US, are conducted under a Food and Drug Administration (FDA) Investigational New Drug (IND) authorization, or involve a drug that is manufactured in the United States or its territories.” Therefore, this study might have missed some trials that are not registered on the website.
The authors reported that 54,728 participants are taking part in 192 clinical trials assessing 158 therapies. Most of the participants are involved in Phase 3 trials. 36 potential treatments are already being assessed in 54 Phase 3 trials, 84 treatments in 89 Phase 2 trials, and 45 treatments in 49 Phase 1 trials.
People at all stages of Alzheimer’s disease development are included in the current trials. However, people at the first stages, who present biomarker or genetic evidence of Alzheimer’s disease but are not cognitively impaired, are represented only in ten trials (5%), while more than half of the trials include people with mild Alzheimer’s disease dementia.
Results coming soon
There are 8 Phase 3 trials (and 29 Phase 2 trials) scheduled for completion in 2026. These include drugs such as metformin, an insulin sensitizer; valiltramiprosate, an amyloid anti-aggregation agent; or Wujia Yizhi, a Chinese herbal therapy, among others. There are also 11 drugs in 14 Phase 3 trials (and 19 drugs in 21 Phase 2 trials) scheduled to be completed before 2026, so reports from those trials might be coming soon.
Dr. Cummings is looking forward to those reports: “Alzheimer’s is no longer an untreatable disease,” he said. “It is now a disease with treatments that successfully interfere in the disease process. In addition to the recently approved therapies, several novel compounds are reading out this year and could increase the number of treatments available for Alzheimer’s. The study of donanemab in cognitive normal people with blood tests positive for very early Alzheimer’s may be read out and will reveal if Alzheimer’s symptoms can be prevented or delayed in those with normal memory function.”
Literature
[1] Cummings, J. L., Zhou, Y., Yang, Y., Zhong, K., Fonseca, J., Osse, A. L., & Cheng, F. (2026). Alzheimer’s disease drug development pipeline: 2026. Alzheimer’s & dementia (New York, N. Y.), 12, e70251.
[2] Brookmeyer, R., Abdalla, N., Kawas, C. H., & Corrada, M. M. (2018). Forecasting the prevalence of preclinical and clinical Alzheimer’s disease in the United States. Alzheimer’s & dementia : the journal of the Alzheimer’s Association, 14(2), 121–129.
[3] Cummings, J. L., Teunissen, C. E., Fiske, B. K., Le Ber, I., Wildsmith, K. R., Schöll, M., Dunn, B., & Scheltens, P. (2025). Biomarker-guided decision making in clinical drug development for neurodegenerative disorders. Nature reviews. Drug discovery, 24(8), 589–609.









